Max
Indoor living soil- the knowledge base
Table of Contents
ToggleHonorary mentions and introduction to the Living Soil Community
I hereby try to compile as good as possible my knowledge collected so far. It is tried to work with the current scientific knowledge, which is constantly changing. Therefore, some information may already be refuted again. Many techniques have also not yet been explicitly determined for cannabis and there is also not much research on multi-cyclic soil systems based on peat. That's why I'm trying to combine anecdotal and scientific knowledge to give the most comprehensive and effective guide to gardening with Living soil for cannabis.
I am already looking forward to lively discussions and also to learn from you. I only ask for a scientific style of argumentation, i.e. just please work with sources and if there are none, argue with data and own experiences.
You can reach me via the Instagram accounts Bavarian_buds/Research-gardens or via the comment function
This article is based on my studies in horticultural science, William Albrecht's soil fertility principle studies, Tad Hussey's podcast "Cannabis Science and Cultivation", Bryant Mason's advice on my soil samples/values, and many people from the forums such as Jonathan Traxler or Nino's Farm from the Indoor Living soil community whose recipes and tips are used as the basis for the sample soil mix included in the third part of the article series.
The plan for this series of articles is to give the basics needed to assemble and grow a "Living Soil" system on your own in three parts.
The first part will be about the general concept, sustainability aspects and the basics of a "good" soil for cannabis. It will break down the three dimensions in which good soil is evaluated (Physical/Chemical/Biological) and highlight the key influencing factors. It can also be considered as a basic knowledge section.
The second part will revolve around the translation of the previously listed quality characteristics into the soil composition of the basic substances, such as peat, via minerals and composts. It follows both an explanation of the test methods and quality characteristics of the individual substances and the soil mixed together to determine from it in process control values to standardize and optimize production as much as possible.
In the third and last part we will go into the culture management of the soil, because this is at least as crucial to success as the right composition of the mix. Finally, I will attach a list and an Excel calculator of my preferred mix for the people who want to start directly with a well thought-out mix.
The concept of Living Soil and the term Modified Growing Mix
Definition
Indoor living soil is currently experiencing a real hype, which is also accompanied by the spread of some misinformation that requires correction. A real living soil, as it was originally called in the permaculture movement in cannabis, describes an outdoor soil in a specific microclimate with local influences of wind, weather and native soil, which is enriched with organic and mineral nutrients such as compost or rock dust, adapted to the plant needs. It has a complete ecosystem that we can only slightly influence, but which, with the right conditions, can have a very positive effect on cultivation. For example, an optimal balance of the system can withstand strong pest pressure through increased bird nesting.
Over time, a biotope of its own develops here and, with proper care, a balance is established. This regulates most problems by itself, such as nutrient deficiencies or drastic pest infestations. We have to create this balance in the indoor system from scratch and can only partially fall back on established biotopes like outside.
Furthermore, we work here with native soil made of sand, silt, clay and rock. This is difficult to implement indoors or has not been experimented with much. The anecdotal evidence I have received so far as to why a peat-based system works better is limited to the high aggregation stability of peat over time, a good pore ratio, a high cation exchange capacity, and the low PH of peat, which allows for high accumulation of cations such as calcium/magnesium. It follows that we don't actually use "soil" at all for "indoor living soil", but a "soilless" mix, as it is known in the English-speaking world. By adding rock meals and other nutrient sources we modify this "soilless" mix even further so the name is changed to modified (soiless) growing mix (MGM) and this is also used as the correct name in the rest of the article.
Indoor living soil is usually referred to a bed or a potting system with at least 60-80L of substrate, which is peat-based and purified by means of minerals / organic fertilizers and used over several runs. Here the goal is to create a diverse and active community of microorganisms, including bacteria, fungi and other beneficial insects. These microorganisms play an important role in plant health and growth by providing essential nutrients (Tegeder and Rentsch 2010)improving soil structure (Rillig and Mummey 2006; Helliwell et al. 2014) and plant resistance to harmful organisms (pathogens).
This means that while it makes sense in some aspects to imitate nature, as in the pest example above, it is also sometimes advisable to use new methods, such as the use of peat. Our goal is to increase yield/quality over inputs. I.e. our goal when introducing new techniques is always to either increase output or minimize the amount of input/environmental damage needed to maximize the total break output/input. This should always be the top priority in the evaluation process.
Why the system can also be sustainable indoors
Some people will now wonder if you do not bring in a lot of pests with all the organic matter. This question is justified and, unfortunately, this is true.
However, this pathogen pressure is easily possible with a well-integrated pathogen management system. This includes prevention through purity, useful insects and in case of need organic sprays, such as Neem oil. However, pest management will be a separate article, otherwise it is beyond the scope.
The benefits, on the other hand, are manifold. With proper, local and conscious purchasing of organic fertilizers, living soil can be cheaper and more sustainable per run. However, great care must be taken in what and where one buys, as often fertilizer materials have long shipping distances or unethical predatory methods as with certain guano manufacturers or rock meals that are mined using open pit mining methods. This must be avoided and always buy as seasonally and locally as possible, otherwise the idea of sustainability is quickly undermined. Optimally, an LCA (life cycle assessment) review for the entire mix has been done beforehand, this gives an overview of all externalities (impacts outside the immediate environment on the entire ecosystem) of the components. The MGM shown later will be CO2/externalities optimized, but not perfect, it will be continuously improved. The best option is to integrate as much as possible into your own cycle. I.e. make your own compost, source aggregate meals from local quarries, and enrich ferments yourself.
Now we talk about sustainability, but use peat as a base, why? There are sustainable extraction practices that improve and balance the CO2 balance, which is negatively affected by the outflowing methane during peat extraction. Care must be taken to ensure that the operation offers a regeneration strategy, as simply balancing CO2 does not do much for the locally degraded ecosystem.
An example of this is the Veriflora® certification. It has good properties such as low storage density (see chapter 2.1.1) to balance humus, has good CEC values (chapter 2.2.2), and is stable over many runs (Burgel et al. 2020). This is especially important if one does not want to remix the soil every 3rd time because the substrate slumps. However, peat cannot provide the pure base substance. Which additives are needed and which values are used for evaluation will be summarized in the following chapters.
Why Living Soil can produce superior plant quality and expression
The topic of quality and its definition fills a separate article, which will appear after this series of articles, and therefore only a small excerpt is shown here
The system is divided into three subject areas. The first category describes the pharmaceutical quality. The remaining evaluation criteria are split into the basic craftsmanship of production/processing and the sensory profile. These are to be evaluated in a pyramid-like manner in the sequence of the main categories (1.1; 1.2; 1.3). That means for an optimal product these must be fulfilled build on each other.

It could be compared to Herzberg's two-factor theory, where pharmaceutical quality can be assigned to the hygiene factors and the other two to the motivational factors. (Becker 2019). This means that the material must always at least correspond to the pharmaceutical qualities before one can even think of evaluating taste or flower processing. In the weighting, a distinction must be made according to the market positioning/stakeholdership of the target demographic up to which category level one must go.
The cornerstones of the pharmaceutical definition must be fulfilled for the use of flowers. Especially in the medical field, compliance with pharmaceutical production standards is a basic requirement. These must be adhered to throughout the entire value chain, from cultivation to packaging.
From GACP to EU-cGxP to HACCP guidelines and the more advanced certifications, there are many ways to meet and exceed the specifications of the European Central Laboratory. These refer to the basic test criteria according to the German Pharmacopoeia (DAB) which consist of purity, identity and active ingredient content. Colloquially, this means: Is the flower free of pesticides, heavy metals, and microbial contamination; is it really cannabis; and does it contain as much active ingredient as stated. (Deutscher Apotheker Verlag 2021).
However, it remains to be seen whether GACP/GMP is absolutely necessary for compliance with these basic qualities; my colleague Lorenz has already dealt with this topic in greater depth. All that can be said is that it is possible to create high-quality, pure products even without a 100-million-euro plant.
This quality level is easy to cover with Living soil, you just have to respect the microbial consortium (chapter 2.3) and work with pharmaceutical purity, so no sprays in flower, work only with beard/hairnets/gloves and submit flower tests (COAs - Certificates of analysis).
The second quality level is the craftsmanship quality. Artisanal qualities include factors that can be objectively assessed in end products and are included in the manufacturing process. These include

- The cultivar (ancestral tree and goodness of selection)
- The cultivation (expression of the cultivar, sustainability)
- The drying
- Trim, treatment and storage of the material
- Consumer-owned ratings such as "White ash".
In particular, the cultivation is of course the decisive factor here. And hereunder the expression of the cultivar, which is the expression of the genetic base material under the influence of the grower and the environment arises. So to speak, a miniterroir, when someone deals with wine. It does not describe that one personally likes the olfactory profile, but that the cut has been well germinated.
Haze, for example: many people hate Haze, but a well-growed Haze, when allowed to go its full 15 weeks and possess fine, deep notes, remains a good Haze in terms of craftsmanship. So it puts its own special characteristics in the foreground. Genetics and the quality of its selection obviously comes as a limiting factor here, as a specific cultivar can only be as well grown as it is genetically capable of being. Therefore, at the beginning of every superior product there is an intensive selection, in order to then tease out the best possible expression (dial in process).
This is possible with all cultivation methods, but faster and easier with Living Soil, because it allows the plant itself to decide which nutrients to take up and when to stop, e.g. to achieve optimal maturation/senescence, more details in chapter 2.2.3 The uptake of nutrients at the roots. To achieve this, of course, the hand tool must sit, so the evaluation of these criteria also falls under the craftsmanship quality.
The final level of quality is then rather subjective and often dependent on the testers in its evaluation as it assesses the sensory quality. While this can also be divided into measurements such as terpene concentration, diversity or THC measurements, in my opinion this does not do justice to the delicate differences of the flower and is based on the approach of science to break everything down. I am more than many a vehement advocate of science, but when it comes to stimulants like wine, cigars, spirits, or even cannabis, I still see man up front. That's why I would prefer a rating system like the Robert Parker points for wine for sensory and overall quality over the measurement-based current systems.
Skilled assessors train for years for these jobs, so it is not so easy to get this skill. Especially in the grey area of cannabis, where intensive black market activity is still needed. This is one of our advantages as non-legal market participants over the new people that are now sneaking in all at once once it becomes legal. So hopefully there will always be a place for people with a sense of quality.
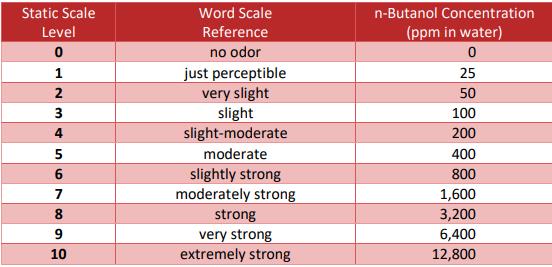
A living, organic system can again be an advantage here, as the decomposition processes in the soil produce hundreds of decomposition products such as caproic acid or carbon-based nutrients, which give the plant completely different possibilities to save energy and form additional defense substances, which we are specifically behind in cannabis. In fact, there are also substances like lactones, which are present in tiny amounts, which are not broken down in most tests and yet have a significant impact on the odor profile. This can be seen quite clearly when comparing cannabis based and synthetic based terpene blends for cartridges for example. This value is given in n-butanol reference values.
Figure 1: Odor intensity classification scale compared to n-butanol. (St. Croix Sensory Inc. 2018)
In summary, Living soil makes it easier to create an optimal expression of the cultivar and to reach the maximum genetic potential. Thus, it facilitates quality levels 2/3, but the first level (pharmaceutical quality) is somewhat more difficult to achieve than with an inert rockwool system, as these are much easier to keep pure.
An exact listing and integrative rating system of the current quality definition follows in the article: What constitutes quality in cannabis.
What is the composition of a good soil
But what all do I have to consider when compiling? First of all, we have to look at the "optimal" MGM. This one does not exist, because every situation, every setup and every growstyle is different. That's why you have to put together the tools and adjusting screws and then adapt the soil mix depending on the situation.
For example, if one has a grow with maximum irradiation (1200umol/s*m2) but no watering system with sensor, a soil with high field capacity (water holding capacity) is advantageous. If one wants more short-term control and can re-fertilize often, the use of quickly available fertilizers in lower concentrations is more advantageous than using a large, slowly available amount of organic fertilizer once.
But such strategies come later, for that first the basics must sit. For this purpose, we will first clarify the most important terms on the subject, which are important in indoor living soil. This will by no means be exhaustive, especially from a scientific point of view. I recommend for this the book (Blume et al. 2010) for a full overview of soil science.
Physical factors
Storage density, grain size and aggregation
Storage density is a measure of the mass of soil in a given volume. It is usually expressed in grams per cubic centimeter (g/cm^3) or pounds per cubic foot (lb/ft^3). Soil storage density is an important physical property that can affect the soil's ability to support plant growth and the movement of water and nutrients through the soil.
There are several factors that can affect the storage density of the soil, including the type of soil, the type and amount of organic matter present, and the composition of the pore profile. Soils with a high percentage of organic matter tend to have a lower storage density because the organic matter takes up space and reduces the density of the soil, this is true for proper soils with clay, in our peat based system the density is pretty much the same as the parent material peat. Similarly, soils with a high percentage of pores tend to have a lower storage density because the pores form voids that are not filled with soil particles.
Storage density can also be influenced by soil management practices. For example, tilling the soil with heavy machinery or rough digging, which breaks up the natural aggregrates, can increase storage density.
Understanding soil storage density can be useful for a number of purposes. In agriculture, for example, knowledge of soil storage density can help farmers optimize irrigation and fertilization practices. It can also be used to predict the movement of water and solutes through the soil, which can be important for predicting the risk of soil erosion and leaching of contaminants. But to read out implications for cultivation, we need more data such as grain size, pore composition, aggregation and the target values.
Grains are defined as the composition of the mineral bodies from primary/secondary minerals, e.g. pumice or silicates in the soil. Aggregates are when these grains or organic particles form a complex together, as in peat. If you have ever opened garden soil, you know what I mean, that is not perfectly homogeneous powder, but should have a nice crumb structure. These "pieces" in the soil are compositions of organic matter, microbiome (bacterial slime), minerals and pore distribution.
Figure 2: Black peat, the substrate with almost perfect structure. (International Peatland Society 2019)
In cannabis cultivation specifically, researchers are in the process of determining what the optimal values for storage density / composition are, but in cultivation itself there is unfortunately little data so far. These are often difficult to determine for living soil substrate mixtures, as both the mixture ratios and the microbial composition are inhomogeneous.
Based on my own tests and advice from Jonathan Traxler, I would recommend a density of 0.4-5g/cm3 or 400-500kg/m3 , but again this is dependent on the objective, we took this value to shift the pore balance towards the center pores to maximize contents in oxygen and water holding capacity while making the soil heavy enough to create an appropriately high Total Exchange Capacity.
These concepts and quality characteristics will be discussed in the next chapter and in sec. 2.2.4 CEC and AEC - Cation and Anion Exchange Capacity and Base Saturation will be explained.
For simple home use, a visual assessment of the structure/aggregation is sufficient by taking a handful, moistening it to saturation and squeezing. It should drip slightly and form a "loose" clump, but this will move apart after a short time. You can also lightly dig up the soil itself by hand and see how big the soil aggregates are. There should be just under 3-4cm3 aggregates/clumps, this is also called crumb structure.
There are studies such as. (Campbell et al. 2021)which look at substrate influence using mineral nutrients and hormones on rooting of cuttings. Here there is a pure series of experiments on the influence of substrates on root rate and root weight. All parameters were positively influenced by low storage density and higher porosity up to a certain point. Too high porosity in the sense that there is a lack of contact between roots and soil leads to loss of turgor, the internal water pressure that keeps the plant upright. (LOACH 1985)
Pore size and distribution
Pores are the spaces between the grains and result from their size/storage, which we considered in the last chapter. They play a crucial role in the physical and chemical properties of the soil, as they can affect their ability to absorb water, air and other substances.
Pore size refers to the diameter of the openings in a material. The size of pores can range from small, such as in clay soil, to much larger, such as in sand. The size of the pores in a material can have a significant effect on its properties.
As the pore diameter decreases, the water is more tightly bound in these pores. The smaller the pore diameter, the higher the suction tension (in cm water column "WS") with which the water is held, corresponding to the conditions in a fine capillary. The pF value (=log cm WS) describes the water retention in the soil in a descriptive form. The higher the pF value, the more "force" it takes to remove the water from the pores. Reference values for this are given in Figure 1: Pore distribution (Blume et al., 2010).. The pF value can be converted into kPA, mbar and other voltage units.
Very fine pores, for example, can only be emptied with the aid of strong negative pressure or heat. This phenomenon is, among other things, responsible for the fact that microorganisms can safely store themselves in the soil even in the case of severe desiccation (sporulation still remains the decisive factor in the survival of microorganisms).
In the table we see the distribution of the pores. We can further divide the coarse pores into wide and narrow coarse pores. Between 10-50mm they are called narrow and >50mm then wide coarse pores. The narrow ones act more like center pores and are a good mix of drainage and water retention. Above 50mm it acts more as pure drainage pores as no water can be held against gravity.
| Pore type | Diameter in mm | Waterpressure | pF-value | Function | Aeration | Plant availability |
| wide | >50 | <0,06 | < 1,8 | Movement of drain and adhesive water |
easy | easy |
| middle | 50-0,2 | 0,06-15 | 1,8 - 4,2 | Water storage (plant available) |
hard | medium-heavy |
| narrow | < 0,2 | >15 | > 4,2 | Dead water (not plant available) |
none | none |
Figure 3: Pore distribution (Blume et al. 2010)
An important function of pores is their role in water retention. Pores that are large enough to hold water, but not so large that the water drains away quickly, are called "effective pores" or central pores. Materials with a high percentage of effective pores are able to hold more water and retain it for a longer period of time. This can be advantageous in areas with low rainfall or drought.
Pores also play a role in the exchange of gases, such as oxygen and carbon dioxide. In soils, for example, pores are important for air movement and the exchange of gases between the soil and the atmosphere. This is crucial for the growth and health of plants, as they depend on the supply of oxygen for respiration and the uptake of nutrients.
For this reason, as mentioned at the beginning, attention must again be paid to the respective application. This determines whether you need more drainage or more water retention capacity, and depending on this, you have to maintain certain mixing ratios for the soil substances in order to obtain corresponding distributions of pores. The goal is always to maintain approximately the profile of peat, so if I now add very dense materials such as biochar or clay/compost mix, I must add aeration materials to the same degree to maintain the balance.
Our basic material peat has excellent properties for a wide range of applications. The pore distribution shows a left-sided deflection, especially in the central pores. This means that the largest volume proportion of the pores is in the plant-available area. Black peat ( c ) is particularly suitable for this purpose.

Figure 4: Pore size distribution of different peat types. (Turunen et al. 2019)
Field capacity and irrigation
Without water, no healthy soil. Without proper watering, the plant's decomposition processes and nutrient supply cannot be maintained. In addition, you can permanently damage your soil if you let it dry for too long.
An important term is the field capacity (FC). It corresponds to the water volume of the soil samples drained to pF 1.8 (=60 cm water column) or taken at corresponding moisture content. This means more or less how much total available water in the soil can be held against gravity in the pores i.e. our maximum available water storage.
According to the classification of pore sizes, the pore range from 0.2 mm to 50 mm is called the usable field capacity (nFK) of a soil, because the contained pore water is available to plants. The water of pores < 0.2 mm is no longer plant-available and is therefore referred to as "dead water" because it would require too much suction from the plant to be used. It is factored out when calculating nFK. The so-called permanent wilting point (PWP) is pF 4.2 or 1000kPA. (Blume et al. 2010). To make the whole thing a bit clearer, you can watch this video
If the PWP is reached during cultivation, the plant dies. However, the point at which wilting begins is different for different plants. Cannabis s., for example, is tolerant and can dry back to high pF values without dying. However, this should only be used to apply targeted stress stimuli, as permanent drought means reduced yield.
Depending on how much % nFK we have filled in the soil, there is a different amount of suction pressure or tension on the root. The drier the soil, the more force the plant has to use to absorb the water.
This suction strength can be measured with a tensiometer and is called matrix potential in soil science. The matrix potential is measured in mbar or kPA. Since little research has been done on this / there are few established values for peat, one must resort to empirical values and anecdotal evidence. These come either from users on the forums or from podcasts such as "Cannabis science and cultivation" by Tad Hussey. One example is an episode from the
The optimum values determined so far also vary from cultivar to cultivar and must finally be determined by the grower himself based on his assessment of the vitality of the plant. The lower the suction tension, the more saturated the soil is with water. In the values we can see that we drive the vegetative phase wetter than, for example, in deliberate stress periods such as the Flower I or Flower III phase in which we want to induce light drought stress.
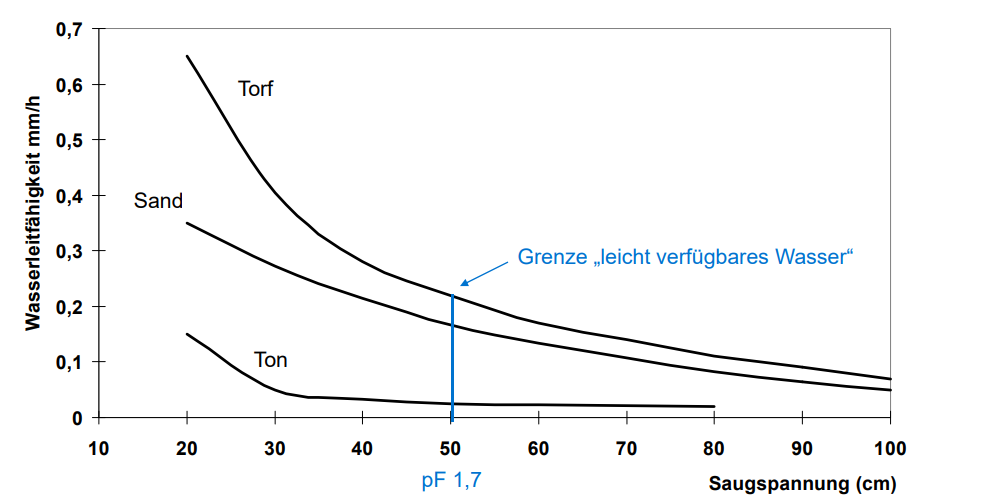
Figure 5: Water holding capacity under suction stress
However, stress should only be applied when there is full control over the system and the plant, because if the plant is already stressed anyway, we do not want to apply more unnecessarily. (Geelen et al. 2019)
There are ways to quantify this stress, but this requires expensive research equipment and is not suitable for the home user. For commercial cultivators, this is enormously worthwhile for establishing cultivation SOPs for the individual cultivars. However, this requires a research manager or consultant who is knowledgeable in this area.
Here are guideline values so far determined by me and the scene for suction tension in indoor, peat-based living soil systems:
| Life phase | Start Veg | Center | End Veg | Flower I: Transition |
Flower II: Bulk | Flower III:
Maturation |
| Mbar Range | 40-50 | 40-60 | 40-80 | 120-160 | 60-100 | 120-160 |
Table 1: mbar ranges for each phase of growth
To optimally control the degree of saturation, you need a good tensiometer as mentioned above. The best not purely commercially available is the Irrometer SR, which is suitable for light soils, such as our peat mixtures. There are also variants with digital output which can be incorporated into an irrigation controller, but to show this in detail is again beyond the scope.
Cheap alternatives can be found at Blumat or similar manufacturers. These are to be used at about 30cm depending on the depth of the bed. It is best to take the center of the bed, but also measure the entire bed at regular intervals to determine any inhomogeneous water distribution and adjust your watering behavior. Now if you want to use a very fancy/high tech term, you could call this watering behavior organic crop steering, although this also involves controlling the environment actually. It is the supreme discipline and we will discuss in chap. 4. procedure and schedule for the use of Living soil discussed.
Chemical factors
Now that we have considered the physical properties as far as possible, we turn to the chemical properties of soil and plant nutrition. First an overview of the 16 nutrients needed for a productive life cycle in plants, then the pH value in focus, followed by the exception principle of nutrients at the root and finally an explanation of the ion exchange principle at surfaces.
Plant nutrients 101
Like all plants, cannabis requires a balance of macro- and micronutrients to grow and develop properly. Macro and micronutrients are needed for a variety of plant processes, including photosynthesis, protein synthesis, and enzyme function. I recommend (Maathuis and Diatloff 2013; Pandey 2018) as reviews on the topic with in-depth physiological explanations.
For detailed information on plant nutrients specific to cannabis, see the research of Dr. Nitrite Bernstein, (Llewellyn et al. 2023) and (Cockson et al. 2019b) recommended, or for practical videos on the topic, the Greengenesgarden YouTube channel.
Reference values, application instructions and recommended concentrations of the nutrients come in the chapter".3. how to test these components of my soil system", because there are some different adjusting screws and you still have to know them to be able to do something with them.
Macronutrients
There are 6 important macronutrients that cannabis plants need for their growth and development. These include nitrogen (N), phosphorus (P), potassium (K), calcium (Ca), magnesium (Mg) and sulfur (S), which are required dry at a minimum of 150g/kg of plant material and are therefore also called macronutrients.
Nitrogen
First to the most important ingredient, as it is the basis for all protein production in the plant, which make up the most functional molecules of the plant. It is therefore integral to growth and biomass. It is also a component of nucleic acids (adenine, guanine, cytosine, thymine, and uracil), which form the basis of DNA information and is required for the production of chlorophyll, which is the functional organelle of photosynthesis. (Pandey 2018)

Figure 6: Nitrogen forms and their use in the plant. (Pandey 2018)
The substance is contained in most chlorophyll-containing tissues of plants and in any amino acid-based substances such as peptides/proteins from e.g. blood/horn/soy. There are a variety of organic compounds that carry nitrogen, but most of it is added to the soil in the form of red composts or worm composts.
These organically bound forms such as humic acid (C9H9NO6) are complex and diverse so an exact list would be too much, but there will be an amendment list still in chapter 2.4.5.
When these substances are digested by microbes or mineralized/available through chemical interaction, most of them are present as ammonium (NH4+) or nitrate (NO3-). Annual plants such as Cannabis s. exhibit ammonium intolerance above a certain, cultivar-specific concentration (Esteban et al. 2016). However, up to this concentration, it can be beneficial to supplement some of the nitrogen via NH4+ , because as seen in Figure 4: Nitrogen uptake and utilization in the plant (Li et al., 2017). this again saves a synthesis step and thus energy / ATP, which the plant does not have to expend.
In this figure, one can also see once again the advantage of direct amino acid application, because these can be directly processed into glumatinic acid and used as a basis for the following synthesis steps. (Pandey 2018)
Figure 7: Nitrogen uptake and utilization in the plant. (Li et al. 2017)
Phosphorus
Another important macronutrient for cannabis plants is phosphorus. It is required for the synthesis of DNA and RNA as well as for the production of energy in the form of ATP (adenosine triphosphate). That is, it is both contained in every cell as a cellular template and is the basis for any final energy source of the cell and is considered the currency for every process that takes place in the plant. (Pandey 2018)
Furthermore, it is the most important component of the membranes, because these are literally phosphorus bilayers, an example is the casparic strip, which acts as a separator and filter in the roots. They are also, along with calcium, the most important biomineral in trichomes and are partly responsible for "white ash" due to their white residues during combustion. (Weigend et al. 2018)

Figure 8: Mineral composition of plant trichomes (Weigend et al. 2018)
It is largely taken up as H2 PO4- and is present in the soil either as inorganic, pure phosphorus or organophosphoric form. Amendments used are mainly rock phosphates such as soft-earth rock phosphate or animal products such as manure, guano or bone meal. (Pandey 2018)
A deficiency results in reduced photosynthetic activity and thus is accompanied by reduced efficiency and lower activity of various coenzymes, making the plant vulnerable to biotic and abiotic stress.
Potassium
Due to the high permeability of plant membranes to K+, plants take up potassium quickly. It is characterized by high mobility in the plant and is translocated to younger tissue.
A close relationship between the water channels and the channels for potassium uptake contributes to the maintenance of plant homeostasis, this uptake mechanism is discussed in more detail in chapter 2.2.3.
It is widely distributed in the cytoplasm and serves to maintain ionic balance and osmoregulation in plant cells by neutralizing insoluble macromolecular anions. Its high concentration in cell vacuoles promotes turgor-controlled development of cell elongation; this is also used, for example, in guard cells of stomata. These are pumped full of K+ via ion channels and water then migrates in through osmosis, causing the guard cells to open.
Potassium deficiency reduces turgor and cell size, resulting in reduced leaf area and stem elongation. The deficiency also damages the cell internal pH regulation and impairs the activity of over 50 enzymes, which again ends in susceptibility to biotic and abiotic stress.
Sulfur also protects against dangerous heavy metal deposition via phytochelatins. Excess heavy metals induce the expression of phytochelatin synthase, which catalyzes the production of the low molecular weight polypeptides known as phytochelatins from glutathione.
This substance is supplemented mainly via red composts, worm composts and minerals such as langbeinite. In KNF, water extracts from potassium-enriching plants such as tobacco are used, i.e. direct addition of potassium.
Calcium
Calcium occurs in plant tissue as Ca2+, Ca-carbonate, Ca-phosphate and Ca-oxalate. Which of these forms accumulate in the soil is of immense importance, since, for example, an accumulation of bi carbonates in the soil makes most nutrients insoluble and thus unavailable to the plant. The uptake of Ca2+ is slow, as it is only absorbed by immature root tips. It is essential for root development as can be seen in the figure, here the root orients itself based on the calcium gradient. (Pandey 2018)
Figure 9: Calcium-dependent root growth. (Carol and Dolan 2002)
Uptake is a passive process inhibited by the presence of K+, Mg2+ and NH4+. A small amount of calcium is maintained in the cytoplasm to avoid unfavorable interactions with other nutrient ions (PO4, Mg2+) and deactivation of enzymes. It is also needed for it to serve as a second messenger. The low cytoplasmic calcium concentration (0.2 M) is maintained by controlling Ca2+ flux across cellular and subcellular membranes. (Pandey 2018)
Membrane integrity and thus the resistance of cell walls is directly dependent on the calcium concentration. Callose, which is enriched at the cell junction shafts (plasmodesmata) as a virus-fighting measure, is also based on calcium. This blocks one of the virus/viroid spreading pathways between plant cells.
A recent discovery is role of Ca2+ as a signal transduction molecule. The perception of stress signals causes calcium channels to briefly open and calcium to be pumped into the cytoplasm, increasing its cytosolic concentration. Which then triggers defense mechanisms in the context of PTI (pattern triggered immunity).
It is an immobile nutrient and must therefore be transported in the irrigation water from the plant through the xylem pathway system to the top of the plant. Deficiencies show up first at the tips.
Magnesium
Magnesium is required in significantly lower amounts than other cations such as calcium. This could be due to high cation competition in absorption and a lack of magnesium transporters in the plasmalemma. Low pH and cations such as K+, NH4+, Ca2+ and Mn2+ inhibit Mg2+ absorption. The rate of absorption is slow.
Magnesium has several different functions. The mobility of magnesium in cells is related to its function. The main function of magnesium comes from its position as the core element of the chlorophyll molecule. This organelle is the cornerstone of photosynthesis and when the central atom is missing, logically, photosynthetic performance is severely compromised (Pandey 2018).
Typical amendments for magnesium supply are algal lime or epsom salt (MgSO4-), which still usefully contains sulfur. Depending on the organic association, however, care must be taken with the use of sulfates, as these are sometimes considered "evil" mineral fertilizers.
Sulfur
Sulfur is present as SO4- i.e. sulphate, the most typical forms are gypsum (calcium sulfate), which is additionally an excellent pH-neutral calcium supplier, and organic compounds such as in composts.
Sulfide reduction in plant metabolism results in the incorporation of sulfide into a variety of different molecules, including the amino acids cysteine and methionine, coenzymes, and secondary metabolites. Cysteine and methionine are both essential components of plant proteins. These amino acids are particularly important because animals and humans are unable to break down sulfur and depend on plants to meet their nutritional needs. This is why an adequate supply of sulfur is important in any plant. Glutathione, the most important detoxification enzyme, is also based on sulfur compounds (cysteine) (Pandey 2018)
Sulfur protects against dangerous heavy metal deposition via phytochelatins. Excess heavy metals induce the expression of phytochelatin synthase, which catalyzes the production of low molecular weight polypeptides known as phytochelatins from glutathione. (Pandey 2018)
In the case of cannabis, the point of increased secondary metabolite synthesis is of particular importance, as these particularly favor VSC (volatile sulfur compounds) accumulation. These substances are responsible for the garlic odor in GMO, for example. However, there are many different ones and they can be most attributed to the gas/garlic notes if you don't differentiate between them. One of the best papers on this topic is. (Oswald et al. 2021).
Figure 10: VSC analysis of Bacio Gelato (Oswald et al. 2021)
Carbon
Not one of the classically valued plant nutrients, but essential is carbon. This is absorbed through the air as CO2 and acts as a source of carbon for glucose production (sugar) in photosynthesis. However, it also has other important roles in the soil. First, every organic matter and many important chelating agents (availability increasing) such as humic acid are carbon based. It serves as a carrier for most nutrients and is the cornerstone of all life.
Storing atmospheric CO2 from the air in the plant and soil after cutting, is the key to reducing greenhouse gases in the atmosphere.
This process is called CO2 sequestration and through careful calculation and compensation areas can thus compensate for the emissions produced (fertilizer/tractor/processing). This is the foundation of sustainable agriculture and the only way to date to truly efficiently remove climate-changing gases from the air.
In addition, this will become a valuable market basis for emissions trading in the future. It is all calculated with a carbon balance and additional points/deductions are given for certain methods. A good review on the methodology was co-published by Prof. Dr. Hülsbergen, from whom I was also briefed on this topic (Küstermann et al., 2008)
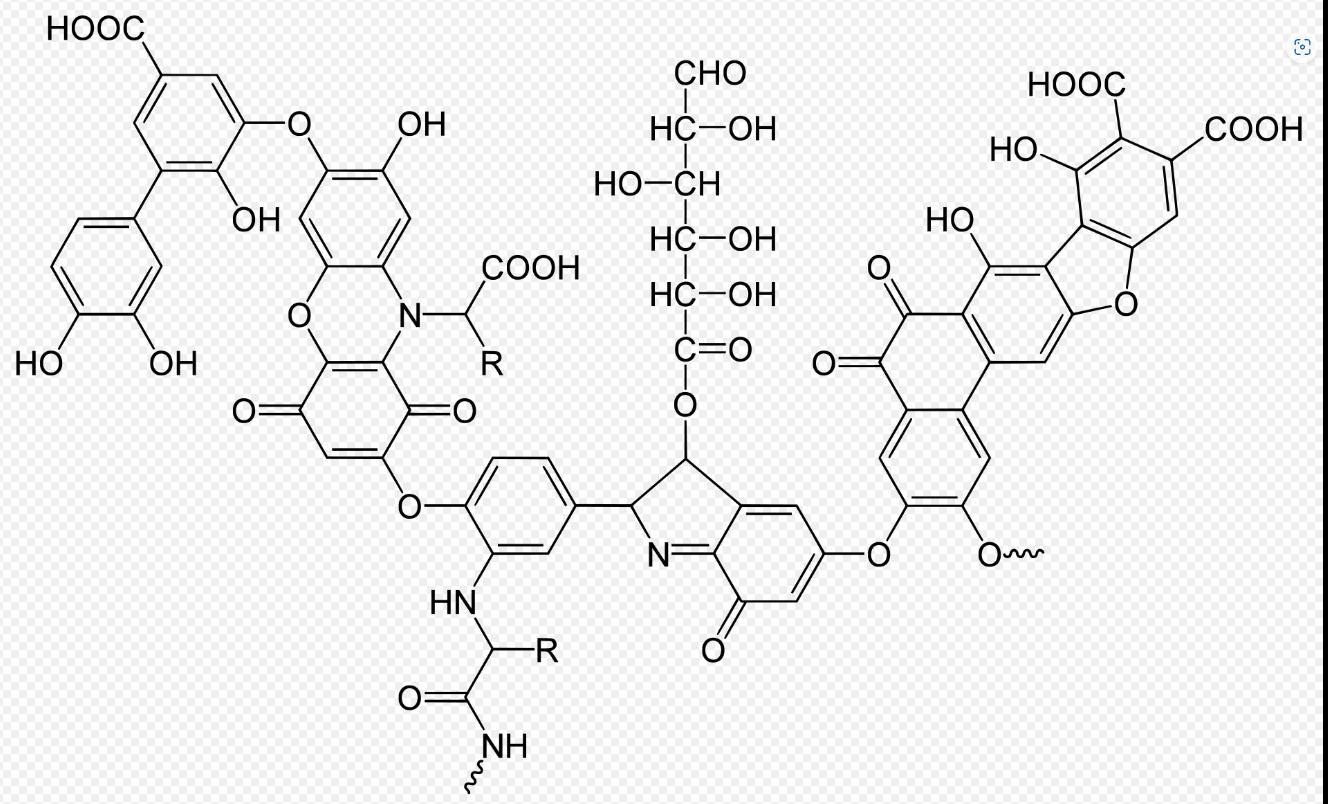
Figure 11: Formula humic acid (Stevenson 1994)
Micronutrients
Anything less than 100 g/kg plant mass dry is called a micronutrient, these are iron, boron, molybdenum, copper, zinc and chlorine. Especially molybdenum, boron and iron are common deficiencies in cannabis so special attention should be paid here (Cockson et al. 2019a). Most micronutrient effects relate to use as a co-factor in enzymes or are involved in the mitochondrial electron transport chain.
Boron takes
Figure 12: Copper-dependent enzymes (Pandey 2018)

Figure 13: Manganese-dependent enzymes (Pandey 2018)
Figure 14: Molybdenum-dependent enzymes. (Pandey 2018)

Figure 15: Various iron-dependent enzymes. (Pandey 2018)
Additives / Strengtheners
Silicone
For plants to maintain their structural integrity, they need the micronutrient silicon. It contributes to the formation of cell walls and the control of plant growth. Together with calcium, this nutrient gives plants their real strength. It is crucial because bud rot can easily penetrate and develop in the large buds, especially under high intensity conditions (1000umol/m2*s, 1200ppm CO2, etc.). In addition, it reduces the need for binding and helps strengthen very heavy branches that might otherwise break. This is especially important for the original GG #4 cut, which naturally forms large inflorescences and flies all over the place. In terms of cannabis, some studies have shown that certain micronutrients may have specific functions in cannabis plants. For example, a study published in the Journal of Plant Nutrition and Soil Science found that zinc deficiency in cannabis plants can lead to reduced growth and cannabinoid production (Landi 1997).
There are other substances that influence plant nutrition but are not directly absorbed as nutrients. They have partly positive, but also negative effects.
Sodium
This element is contained in most nutrient carriers such as kelp or mussel shells, which come from the sea. It is not directly needed by the plant and an accumulation is even harmful for the plant, because the substance displaces the other nutrient cations with its positive charge.
Chlorine
Maintenance of turgor and osmoregulation is an essential function of chlorine in plants. The osmoregulatory functions of chlorine include its involvement in turgor-regulated cell development and stomatal function. Chlorine accumulates in large amounts in root and shoot tips, where it is involved in turgor-driven development of cell processes. Nevertheless, too high levels should be avoided to limit the oxidizing effect on bacteria and micro life.
In most cases, enough chlorine is introduced into the system via composts or the water so that it does not have to be supplemented extra.
Conversion formulas and chemical nutrient forms
It should be noted that in the Amendments, the contents are mostly in their bound form such as P O25 .
The conversion factor between P2O5 and P depends on the molecular weight of the respective compound.
The molecular weight of P2O5 is 141.94 g/mol, and it contains two phosphorus atoms.
The molecular weight of P is 30.97 g/mol.
To convert P2O5 to P, multiply the P2O5 value by 0.4364 (or divide by 2.29).
This means that 1 gram of P2O5 contains 0.4364 grams of P.
Therefore, to convert from P2O5 to P, you can use the following formula:
P = (P2O5 * 0.4364)
Or
P2O5 = (P / 0.4364)
Here is a list of all plant available forms of nutrients, you can calculate the pure amount as in the example above.
- Nitrogen (N)
- Nitrate (NO3-)
- Ammonium (NH4+)
- Phosphorus (P)
- Orthophosphate (H2PO4- and HPO42-)
- Mono- and diesters of phosphoric acid (organic P)
- Potassium (K)
- Potassium ions (K+)
- Calcium (Ca)
- Calcium ions (Ca2+)
- Magnesium (Mg)
- Magnesium ions (Mg2+)
- Sulfur (S)
- Sulfate (SO42-)
- Elemental sulfur (S) in oxidized soils.
- Iron (Fe)
- Iron (Fe2+)
- Manganese (Mn)
- Manganese ions (Mn2+)
- Zinc (Zn)
- Zinc ions (Zn2+)
- Copper (Cu)
- Copper ions (Cu2+)
- Boron (B)
- Borax (Na2[B4O5(OH)4])
- Chlorine (Cl)
- Chloride (Cl-)
- Molybdenum (Mo)
Deficiencies in these elements result in losses in general biomass and decreases in harvest index. To quantify the impact have (Llewellyn et al. 2023) laid out a very extensive study that measured the impact.
Figure 16: Fresh weight (FW) of detached aboveground tissue, harvest index (inflorescence FW/stalks and leaves FW), and dry weight (DW) of roots from each nutrient treatment. (Llewellyn et al. 2023)
Figure 17: Change in cannabinoid composition under specific nutrient deficiencies. (Llewellyn et al. 2023)
However, these losses are specific to the location of the organ, as plant nutrients are either mobile or immobile within the plant itself.
The concept of mobile and immobile nutrients refers to how easily these nutrients can move within a plant after being taken up by the roots. Mobile nutrients can easily move from older to younger plant tissues, while immobile nutrients remain in the tissues where they were originally taken up.
Mobile nutrients include nitrogen (N), phosphorus (P) and potassium (K), which are important macronutrients for plant growth. These nutrients can be easily transported within the plant via the phloem, a specialized vascular tissue that transports nutrients and sugars through the plant.
Immobile nutrients, on the other hand, include calcium (Ca), sulfur (S) and many micronutrients such as iron (Fe) and zinc (Zn). These nutrients are not easily transported within the plant and remain in the tissues where they were originally taken up.
Nutrient mobility can have important effects on plant growth and development. For example, if a plant is deficient in a mobile nutrient such as nitrogen, it will transfer nitrogen from older leaves to younger leaves to maintain growth. This can cause older leaves to turn yellow or brown because they no longer contain nitrogen.
In contrast, a deficiency of an immobile nutrient such as calcium can result in stunted growth or malformations in affected tissues because the nutrient cannot be readily transported to other parts of the plant to support growth.
This is immensely important in order to be able to isolate deficiencies at an early stage and to initiate appropriate re-fertilization. One of the best papers in the field, and one of the first in medicinal/current cultivars, is that of (Cockson et al. 2019b).
The PH value and the influence on the ionic form of the nutrients
To ensure maximum growth and development, it is important to ensure that cannabis plants receive the right mix of each of these 16 nutrients. Cannabis plants have different nutrient needs depending on their stage of development, the type and pH of their soil, and the presence of other nutrients. To determine the nutrient content of their soil and adjust their fertilization practices accordingly, growers can use soil tests and plant tissue analysis because the charge and shape of nutrients is affected by the milleu.
In most cases, the nutrients mentioned above are not present in neutral form or are already ions themselves. The charges play an important role in the uptake of nutrients by the roots. Up to now, the uptake capacity of nutrients has been determined on the basis of the table of (Peterson 1982) was used.

Figure 18: Availability nutrients in a peat-based and enriched system. (Peterson 1982)
Recent studies by (Hartemink and Barrow 2023) however, show that this is not so easily applicable. There are still so many interactions at play, such as the various effects of microbial community changes due to exudates, that one can only estimate these PH ranges for inert systems. Even this is still imprecise; influences of root exudates on PH and mineralization rates would also have to be determined to make accurate statements. But this is already beyond the scope of what is possible in home cultivation. That is, especially in living soil systems, neither the PH of the water nor that of the soil can provide accurate information about nutrients. (Hartemink and Barrow 2023).
Roughly, this is nevertheless to be considered in cultivation. Because a soil PH of e.g. > 9, suffers quite certainly from a too high accumulation of calcium carbonate or similar substances. Here, the uptake of nutrients is severely restricted, especially that of micronutrients, which are essential for the regulation of almost all processes and for enzymes. For the pure mineral availability of nutrients, 5.5-5.8 is optimal as it is also used in mineral, commercial cultivation.
However, a pH below 6.0 then becomes problematic again for the microbiome, as below 6.0 it partially switches to an anaerobic, heterofermentative milieu, which we want in certain phases (Stretch/Flower I), but not in most phases. Here we want to maintain a pH of 6.0-6.8, as this is where most of the beneficial microorganisms live, cf. 2.3.1 Micro Flora.
If you now want to create an optimal, cultivar-specific recipe, you can set the p.H. higher or lower depending on the nutrient requirements of the cultivar. My personal sweet spot is 6.5 in the vegetative phase, which then decreases to 6.3 in the course of flowering. However, as mentioned above, this is only an estimate, since microbial life has to be taken into account. In the next chapter, we will also see that the plant/microbe interaction has a considerable influence via the release of sugars and the subsequential propagation of special microbes by the plant. Thus, the plant controls most of the processes and we give it more or less only the tools to be able to develop independently, optimally.
The uptake of nutrients at the roots
Plants absorb nutrients through their roots in the form of ions. These ions arrive at the root via 3 mechanisms. These are interception, mass flow and microbial interaction, the first of which refers to root penetration and subsequential uptake. The second mechanism (mass flow/mass flux) refers to the transport by means of water, which directly flushes ions into the root, this can also be disadvantageous if a too high dissolved ion concentration (too high EC value) is reached in the nutrient solution of the soil. Then the plant can no longer defend itself against the nutrient, so to speak, and is overfertilized / force-fed. Unfortunately, this does not result in a better tasting end product as in the case of foie gras, but rather the opposite and it "burns" the plant. That's why we have to test in tests (chap. 3. how to test these components of my soil system) we always have to pay attention to the mineralization rate, this is determined by comparing stock tests (Merlich III) with solution tests (saturated paste) and measuring the % solution/unit of time. This is of course only an estimate, as we do not have real time data on the mineralization rate. If this is too high, the EC value of our soil solution will also be too high and the plants will die.
The third way nutrients get into the plants is the interaction of microbes (mostly arbuscular endomychoraceae) with the roots. Exact mechanisms and preferred nutrients for this uptake mechanism are discussed in chap. 2.3.2.2 Fungi (PGPFs) will be discussed.
The movement of ions in and out of plant cells during interception is regulated by the concentration gradient, which is the difference in ion concentration between the inside and the outside of the cell. When the ion concentration on the outside of the cell is higher than on the inside, ions flow into the cell to equalize the concentration. This movement of ions into and out of the cell is facilitated by ion transporters, which are proteins that help move ions across the cell membrane. (Wang et al. 2006)
In addition, more highly charged substances such as Mg2+ are absorbed more strongly than K+ with one charge. This can lead to a displacement effect at the root if the concentration of individual substances is too high, since only the strongly charged Mg2+ are taken up, but the weak K+ firstly cannot get through and secondly are repelled by the Mg2+ since they have the same charge. This shows the immense importance of balance in the system, MGMs are like ballet and not like a strongman competition, harmony counts and not much helps much. Especially the cations need to be kept in mind, because while the microbes can balance a lot, they also can't balance everything. We create, as mentioned before, the perfect toolbox for the plant and if there are too many nails, the plant will not find any more screws, to put it a little more vividly. More about this in chapter 2.2.4 CEC and AEC - Cation and Anion Exchange Capacity and Base Saturation
And here again a scheme for nutrient uptake based on a root cross section
Figure 19: Schematic of nutrient uptake based on a root cross-section. (Wang et al. 2006)
We see here on the left side the passive transport. This is mainly for nitrogen derivatives and other charged nutrients. The plant does not have to do any "work" here, as it flows in along the natural H+ gradient. I.e. the charge within the root fits so that the nutrients can be absorbed and the anion/cation balance still fits.
Active transport is used when the work-free uptake path is blocked. Either H+ is expelled from the root during cation uptake, because otherwise there is too much + charge in the root. These channels are called antiporters.
For anion uptake against the natural gradient, a symporter channel is used. This channel packs the - charged particle together with an H+ atom in order to keep the balance and not to disturb the charge equilibrium. In summary, the plant must always maintain a charge balance and exchanges either equal or different charges with the soil. The plant can also control the internal charge composition, but of course always prefers to do as little work as possible itself.
For this reason, there are also higher-molecular, organic compounds such as amino acids, which can be absorbed and thus cost the plant less build-up energy (ATP) than low-molecular nitrate. However, these must be applied directly through the leaf, because in the soil they are pre-digested by the microbes and are then pure nitrate again, which, then must first be built up again. I.e. amino acid fertilization via the soil is relatively useless for the plant itself, but you can use it as food for microbes, but I would not take expensive pure aminos, but compost or fresh green cuttings. Here again an overview of the nitrogen cycle of the plant, which starts with glutamine from nitrate and thus forms the basis for all amino acids on this planet. There are almost no other organisms that can do this step. Without them, there would also be no humans or animals from meat (Tegeder and Rentsch 2010). Which transporters are responsible for which function, we see in Figure 10&11.
Figure 20: transporter modus operandi pt1 (Sasse et al. 2018)
Figure 21: Transporter modus operandi pt 2 (Sasse et al. 2018)
We have already looked at the uptake of plant nutrients at the chemical level, but now we are still missing the interaction of plant and microbes mentioned above. Plants can excrete certain polysaccharides / monosaccharides (multiple / simple sugars) from their roots, which then serve as specific food for specified microbes. I.e. the plant can independently, if it has all the necessary substances / is healthy, control the composition of the microbiome.
If the plant now requires more phosphorus, for example, sugars are exported to the rhizosphere by means of the SWEETS or MFS transporters, as can be seen in Figure 10. In addition, Arabidopsis plants (model organism) deficient in phosphate produced more coumarin and oligolignol, poplar (Populus tremula) treated with heavy metals produced more organic acid, and wheat deficient in zinc produced more phytosiderophores. Differential exudation is one possible pathway by which plants can alter their interactions with microorganisms, as shown by the relationship between exudation patterns and rhizobiome variation in eight Arabidopsis lines. With this and Figures 10&11, we see that plants not only exude these sugars for microbial interaction but can also exude organic acids (elimination nutrients & pH regulation), amino acids and other chemicals. This has hundreds of regulatory effects. The best systematic review on the subject can be found at. (Sasse et al. 2018)
More detailed information on the organisms is presented in chap. 2.3.1 Micro Flora is dealt with.
Up to 20% of the plant's total sugars produced (photosynthesis/Calvin cycle) are spent on these interactions when needed. (Sasse et al. 2018) This number must be reduced so that we cover the basic needs with mineral nutrients, which do not require microbial breakdown, and let the microbes cover the specific characteristics of the cultivars. I.e., balance is again of paramount importance. We do not want to have everything produced by the microbes, but only the fine tuning of the nutrients. Basic needs are provided by minerals/mineralized nutrients. This is also the reason for the optimal expression of the
CEC and AEC - Cation and Anion Exchange Capacity and Base Saturation
Now we have looked at both the soil water supply and what nutrients can dissolve in that water. Now we only need information about the nutrient storage capacity of our soil.
The cation exchange capacity (CEC) describes the ability of a material to exchange positive charge carriers (cations). The counterpart to this is the anion exchange capacity (AEC), which describes the ability of a material to exchange negative charge carriers (anions).
The CEC and AEC of peat are usually measured in milliequivalents per gram (meq/g). The CEC and AEC of peat depend on several factors, such as the chemical composition of the peat, the size and shape of the peat particles, and the conditions under which the exchange takes place.
Unlike in the grown soil of sand, silt and clay, our peat-based system without amendments is still quite meager in terms of plant nutrient storage capacity. In normal soil, there are clay and other mineral compounds with positive/negative charged surface for this purpose.
Figure 22: Cation exchange capacity of different materials (Andrews 2013)
Peat contains many quartz sand grains and organic components, both of which are electrically charged. The organic components have a negative charge, while the quartz sand grains have a positive charge. When peat comes into contact with water, the cations and anions from the solution can bind to the electrically charged particles in the peat. This exchange process is called adsorption.
Soil additives such as minerals (zeolite, langbeinite, etc.) as well as organic substances such as humus/compost improve the storage capacity of the soil and are therefore added. They therefore contribute not only to the filling per se i.e. the pure nutrients, but also to the overall storage capacity of the soil.
The percentage of filling of CEC/AEC is called base saturation and depends on the respective cations/anions already mentioned in 2.2.1 and on the PH milieu. (Blume et al. 2010)
There is no "optimal" value for the cation exchange capacity (CEC) of peat in cannabis growing systems, as the ideal CEC depends on many factors, including the type of cannabis plant, the nutrient requirements of the plant, and the conditions of cultivation. In general, a higher CEC is considered beneficial because it can help ensure that more nutrients are absorbed by the plants. However, too high a CEC can also result in too many nutrients being locked up in the plant, which can lead to a nutrient deficiency
It is measured in meq/100g (milliequivalents/100g). The individual substances and their CEC can be found in chapter 2.4. These milliequivalents can be converted into mg/kg with the respective conversion factor of the specific elements:
Table 2: CEC conversion factors (Local Land Services (NSW Government) 2020)
| Cation | mg/kg or ppm(A) | Conversion factor (B) | meq/100g (A/B) |
| Ca2+ | 582 | 200 | 2,91 |
| Mg2+ | 168 | 122 | 1,38 |
| K+ | 59 | 390 | 0,15 |
| Na+ | 27 | 230 | 0,12 |
To calculate the full CEC, the meq/100g of aluminum and the free hydrogen must still be added. However, mostly attention is paid to the four main components, since free hydrogen occurs in small amounts (at appropriate pH of 6.2-6.8) and in a healthy soil the aluminum should not be present in high amounts either way. (Local Land Services (NSW Government) 2020)
The CEC can be filled with the main cations in Table 1 in various ratios. The optimal values used today are mainly based on the works of (ALBRECHT 1959]. Different crops and different setups must of course always be slightly adjusted according to conditions. For example, heavy soils need more calcium than magnesium in relation to lighter soils. This is due to the coarser structure of calcium and the resulting additional pores.
Three reference values are used, once from (Logan Labs 2023)once from (ALBRECHT 1959) and once from AgPHD on Youtube, which I can only recommend to everyone.
Table 3: Comparison of different building saturation reference values
| Cation/Method | Albrecht | Logan Labs | AgPHD |
| Ca2+ | 68-70% | 60-70% | 65-80% |
| Mg2+ | 10-12% | 10-20% | 12-20% |
| K+ | 4% | 2-5% | 4-8% |
| Na+ | As low as possible | As low as possible | As low as possible |
Now, to convert the optimal values into PPM nutrient, use the formula
CEC x nutrient specific value x selected % base saturation
25 (CEC for example soil) x 400 (value for calcium) x 70% (Albrecht %rate for calcium)
= 7000kg/ha = 0.7 kg/m² = 700g/m2 calcium per m2 bed
7000kg/ha / 2.24 = 3125mg/kg soil (Local Land Services (NSW Government) 2020) l
=> optimum value
An example of how to arrive at the required fertilization quantity with a real soil test can be found in chapter 3.2.5 Cations and the base saturation
The calculated value is unfortunately in kg/ha so difficult to adapt to indoor beds. However, you can roughly calculate and estimate the A horizon with 0.5m and then calculate the ha down to m2 bed.
The nutrient specific values of the cations are:
Calcium: 400
Magnesium: 240
Potassium: 780 (Logan Labs 2023)
Biological factors / The Soil food Web
As "Soil Food Web" is usually described in the popular forums the complex ecosystem in the soil, which consists of different species of microorganisms, fungi, worms, insects and other animals. This term was popularized and spread by Dr. Elaine Ingham, and current science is also turning and giving more and more importance to the Soil food web.
Living organisms interact with each other to form a dynamic network of relationships in which nutrients and energy are diverted in a variety of ways. The Soil Food Web is considered the vitality of the soil and is important for soil health, and through the soil affects nutrient supply, resistance to pests, water uptake and the possibility of plant growth.
The cycling aspect becomes even more useful to us as genera such as bacteria are eaten and digested by larger creatures such as the protozoa or worms, which then provides the nutrients mineralized for the plant when excreted. When digested by worms, additional nutrients are even chelated by humic acids.
This cycle goes through all stages until it finally includes macro creatures like birds, which we unfortunately do not have in indoor cultivation, but these are simulated by digging up / mechanically killing the soil / its creatures, so it is almost a complete cycle. 
Figure 23: The Soil Food Web - Scheme. (Natural Resources Conservation Service 2023)
We now take a close look at individual parts and clarify what use they have in the horticultural sector.
We can further divide these organisms into micro fauna and micro flora, where fauna is the current animal diversity and flora is the current plant diversity.
Micro Flora
Bacteria
This smallest participant of the microfauna is the bacterium. There are an enormous number of strains of them (this is the correct use of strain, cannabis has varieties) and many have beneficial properties for plant growth. This group is called PGPR (plant growth promoting) bacteria.
These have direct and indirect effects on the plant, here is a list of the processes they favor. Those interested in more detail can get a rough overview in the book series Teaming with microbes (Lowenfels and Lewis 2016) or get a very deep, accurate one in the review (Goswami et al. 2016). The review by Goswami et al. contains all the references needed for the individual proofs, actually these should be listed individually for a perfect citation style, but I don't have the time, please just look in the respective chapters.
Direct mechanisms:
- Biological nitrogen fixation
Biological nitrogen fixation is the process by which atmospheric nitrogen gas (N2) is converted to an ammonium form (NH4+) that can be used by plants. Since nitrogen is an essential component of plant growth but is not readily available in most soils, this process is critical.
Nitrogen fixation is the method used by PGPR bacteria to fix nitrogen, including Rhizobium spp. and Bradyrhizobium spp. This process takes place in specialized organs known as nodules, which develop on the roots of some plants, such as legumes. With the help of an enzyme, nitrogenase, the PGPR bacteria in these nodules convert nitrogen gas into a usable form.
- Solution of the phosphorus that is not available to plants.
The process of phosphate solubilization makes phosphate (PO4 3-) more soluble and thus more available to plants. Although phosphate is an important plant nutrient, it is often present in soils in an inaccessible form, e.g. as inorganic phosphate.
Inorganic phosphates can be dissolved by PGPR bacteria such as Bacillus and Pseudomonas species by producing organic acids such as citric and malic acids. The bacteria release these organic acids, which can chelate with the inorganic phosphates and increase their solubility in water. This allows the plants to absorb the phosphates more easily and use them for their growth and development.
Pseudomonas and Bacillus species that dissolve organic phosphorus are able to remove phosphorus from phytate (Found in manure and compost) and other phospholipids. The inorganic phosphates are then available to the plants after the bacteria have degraded the organic forms of phosphorus.
Again, this is solved by the excretion of citric and malic acid compounds. Other mechanisms such as proton excretion through ammonium (NH4+) accumulation is also possible, but there is only a limited difference in the way the methods work, as the organic acids also only act as proton donors. (Park et al. 2009)
- Phytohormones production
Phytohormones are also synthesized by PGPRs. These serve as plant growth regulators, or PGRs, which help control plant growth. Depending on the desired effect, a different type of PGPR can either be grown naturally and supplemented or isolated in the laboratory and added. This can be used, for example, to induce a compressive effect by cytokinins.
The following is a list of phytohormones produced by various PGPRs:
- Auxins: PGPRs such as Pseudomonas fluorescens, Pseudomonas putida, and Rhizobium leguminosarum have been found to produce auxins responsible for cell elongation and differentiation. They also promote lateral root development and fruit ripening.
- Cytokinins: PGPR such as Bacillus amyloliquefaciens, Bacillus subtilis and Azospirillum brasilense have been found to produce cytokinins, which are responsible for cell division and the development of roots, shoots and leaves. They also promote the development of lateral roots, the ripening of fruits and the development of chloroplasts.
- Gibberellins: PGPR such as Pseudomonas fluorescens, Pseudomonas putida, and Rhizobium leguminosarum have been found to produce gibberellins, which are responsible for cell elongation and differentiation. They also promote seed germination and shoot growth.
- Abscisic acid: Some PGPR such as Azospirillum brasilense produce abscisic acid, which is responsible for triggering dormancy and seed maturation and promotes plant tolerance to environmental stress.
- Jasmonates: Some PGPR such as Pseudomonas syringae have been reported to produce jasmonates, which are phytohormones involved in plant defense response.
Indirect mechanisms:
- Siderophore production
Siderophores are tiny, iron-binding substances formed by microorganisms such as specific bacteria that help them absorb iron from their environment. To help absorb iron from the soil, several plant growth promoting rhizobacteria (PGPR) are capable of forming siderophores.
- Pyoverdins, a substance produced by Pseudomonas spp. that can dissolve iron from iron-containing minerals such as iron oxides and hydroxides,
- The production of bacillibactin by Bacillus spp. allows bacteria to chelate and utilize iron.
By forming siderophores that can also dissolve other micronutrients from the soil, some PGPR have also been shown to affect the availability of other micronutrients such as zinc and manganese, which are comparable to iron in terms of solubility and uptake by plants. (Płociniczak et al. 2013)
- Chitinase and glucanase production by PGPR.
Chitinase and glucanase are both types of cell wall degrading enzymes produced by plant growth promoting rhizobacteria (PGPR). Chitinase is an enzyme that degrades the chitin component of fungal cell walls, while glucanase degrades the beta-glucans found in the cell walls of various organisms, including fungi and bacteria.
An example of a bacterium that produces chitinase is the soil-dwelling Pseudomonas aeruginosa, which has been shown to produce chitinase when growing in the presence of chitin. Another example is Bacillus subtilis, which is known to produce both chitinase and glucanase.
When PGPR produce chitinase and glucanase, they can degrade the cell walls of pathogens such as the fungus Fusarium and other potential plant pathogens that have chitin or beta-glucans in their cell walls. This can lead to suppression of pathogenic infections in the roots and rhizosphere of plants.
In addition, these enzymes can also degrade the cell walls of non-pathogenic microorganisms and organic matter in the soil. This allows plant-available nutrients such as phosphorus, nitrogen and other micronutrients to be released from the degraded cell walls, which can be utilized by the plants.
Furthermore chitin fragments produced by chitinase can induce systemic acquired resistance (SAR) in plants, which is an important mechanism of plant immunity to pathogenic infections and receives its own chapter. (Chet et al. 1990)
Overall, the production of chitinase and glucanase by PGPR is a key mechanism by which these bacteria can promote plant growth and health by increasing the availability of nutrients and suppressing pathogenic infections.
- Antibiotic production through PGPR
In a process known as "antibiotic production by plant growth-promoting rhizobacteria" (PGPR), certain bacteria produce substances with antibacterial properties. These substances can promote plant development and nutrient uptake while helping to protect plants from disease.
Biosynthesis and secretion are the two main processes by which PGPR antibiotics can be generated. In secretion, bacteria release the antibiotic into the environment, whereas in biosynthesis, bacteria actively synthesize the antibiotic in their own cells.
The bacterium Streptomyces griseus is an example of a PGPR that synthesizes antibiotics. Numerous antibiotics, such as griseofulvin and streptomycin, are produced by this bacterium. Another example is the bacterium Pseudomonas fluorescens, which is known to produce the antibiotic chemicals phenazine-1-carboxamide (PCN) and pyoluteorin (Plt). (Hammer et al. 1997)
- Induced systemic resistance
Induced systemic resistance (ISR) is a phenomenon in which the application of certain microbes, such as plant growth promoting rhizobacteria (PGPR), can activate a plant's natural defense mechanisms, resulting in increased resistance to pathogens. This is an important strategy for disease control in crops, as it allows plants to be protected without the use of chemical pesticides.
There are several mechanisms by which PGPR can induce systemic resistance in plants. One of the most important mechanisms is the production of signaling molecules known as elicitors of systemic acquired resistance (SAR), such as salicylic acid (SA) and jasmonic acid (JA). These molecules are produced by PGPR and can activate the plant's SAR response, resulting in increased resistance to pathogens.
Another mechanism is the production of enzymes such as chitinases, beta-1,3-glucanases and peroxidases. These enzymes are known to break down the cell walls of pathogens, making it difficult for them to infect the plant. In addition, the chitin fragments produced during the disruption are recognized as PAMP (pathogen associated molecular pattern). This is a form of marker by which the plant initiates its active defense mechanisms (PTI = pattern triggered immunity). (Couto and Zipfel 2016)
An example of a PGPR strain known to induce systemic resistance by triggering SAR is the bacterium Pseudomonas aeruginosa. This bacterium produces a number of molecules that can activate SAR, including SA and JA. Another example of a PGPR strain known to induce systemic resistance by producing enzymes is Bacillus subtilis, which produces chitinases, beta-1,3-glucanases, and peroxidases. (Goswami et al. 2016)
- PGPR modulates plant stress markers under abiotic stress.
Abiotic stress is any stress to which a plant is exposed that is not biological in nature, such as drought, high salinity, or harsh temperatures. These stressors can alter the expression of certain genes, resulting in stress indicators that can be used to measure the extent of stress.
Under abiotic stress conditions, PGPR can alter plant stress indicators through a number of different methods. The formation of molecules that promote plant growth, such as indoleacetic acid (IAA) and cytokinins, is one of the key processes. These substances can help mitigate the effects of stress by promoting plant growth and protecting the plant from stress-related damage.
Another mechanism is the production of enzymes such as dehydrin, proline, and the antioxidant enzyme superoxide dismutase (SOD), which can help protect the plant from stress-related damage by stabilizing the cell membrane, preventing protein denaturation, and reducing the production of reactive oxygen species (ROS) in the plant.
An example of a PGPR strain known to modulate plant stress markers through the production of plant growth-promoting compounds is the bacterium Bacillus subtilis. This bacterium is known to produce IAA and cytokinins that can promote plant growth and protect the plant from stress-related damage. Another example is the bacterium Pseudomonas putida, which is known to produce the enzymes dehydrin and proline, which can help protect the plant from stress-related damage by stabilizing the cell membrane, preventing protein denaturation, and reducing the production of ROS in the plant (Niu et al. 2012; Goswami et al. 2016; Rasool et al. 2013).
- Preparation of 1-aminocyclopropane-1-carboxylic acid deaminase.
1-Aminocyclopropane-1-carboxylic acid (ACC) deaminase is an enzyme that converts ACC, a precursor of the plant hormone ethylene, into ammonia and alpha-ketobutyrate.
The production of ACC deaminase by PGPRs is a well-documented mechanism in the regulation of plant growth and stress responses. ACC deaminase breaks down ACC, a substrate of the plant hormone ethylene, into ammonia and alpha-ketobutyrate. This effectively lowers ethylene levels. Ethylene is a hormone known to have an inhibitory effect on plant growth and is also associated with stress responses. (Etesami et al. 2015)
An example of PGPR that produce ACC deaminase is Pseudomonas spp. This group of bacteria produces ACC deaminase and has been shown to have growth-promoting effects on a variety of plant species.
Another example is the bacterium Burkholderia spp. which reportedly produces ACC deaminase and has been shown to promote growth and improve plant tolerance to various abiotic stresses such as drought and high salt concentrations.
Archae
Microorganisms with a single cell belong to the archaea domain. They are a diverse group of living organisms that differ both chemically and genetically from bacteria and eukaryotes (the other two domains of life).
Archaea are found in a variety of environments, including harsh environments such as salt pans, hot springs, and deep-sea hydrothermal vents. They are also found in more temperate environments such as the soil and the sea.
Both bacteria and archaea are important components of the soil microflora and have a significant impact on soil processes that influence plant growth and health. Both types of microbes can influence plant growth by decomposing organic matter and releasing nutrients that can be taken up by plants, promoting the development of favorable soil structure, and acting as a barrier against pathogens.
One of the main differences between archaea and bacteria is the process of energy production. While many bacteria use oxygen in respiration to produce energy, archaea are often anaerobic and produce energy through a process known as methanogenesis. Methane is the result of the metabolism of methanogens, which are archaea. This can significantly affect the carbon cycle in soils, and methanogenesis may even contribute to increased soil fertility, according to some research.
The ways in which bacteria and archaea interact with plants also differ. While some bacteria have been shown to have direct reciprocal relationships with plants, for example by fixing nitrogen in nodules on the roots of legumes or by supporting plant growth through the production of appropriate chemicals, this is generally not the case for archaea. Archaea may also be involved in the development of biofilms on plant roots that can help protect plants from disease. Some archaea have been shown to produce volatile organic chemicals such as methanol or formaldehyde, which can stimulate plant growth by improving nutrient availability.
They are also involved in the sulfur cycle. They oxidize sulfur and produce sulfuric acid with it, which can dissolve minerals from stones. Calcium, for example, can be converted to calcium sulfate if calcium-containing compounds are available. This substance is commonly referred to as gypsum and provides both good aeration due to its pore structure and a source of nutrients when it decomposes. (Lin et al. 2010; Naitam and Kaushik 2021)
Micro Fauna
Before we go into the exact tasks of soil organisms, we need to establish a classification according to their tasks.
The so-called necrotrophs, feed on dead or decaying substances. They are critical to the decomposition process that helps return nutrients to the ecosystem. They actively kill the plant to get at the dead tissue.
Microbes that feed on living things are called biotrophs. They often form symbiotic partnerships with plants, animals and other species, each receiving a benefit in exchange for the other.
Depending on the resource supply, facultative microorganisms can exist in either living or dead substances. Depending on the environment, they can occur as necrotrophs or biotrophs.
Microorganisms that feed only on substances that are already dead or decaying are called saprophytic microorganisms. They are critical to the decomposition process that helps return nutrients to the ecosystem. Bacteria, fungi, and various species of algae are some examples of saprophytic microorganisms. (Agrios 2005)
Algae
The influence of algae and their produced substances in the soil are already known since the 1960s and conditional researches have already been established. The review by (Dmytryk and Chojnacka 2018) gives a very good overview of their suitability as producers of biostimulants and use as plant fortifiers.
Aquatic organisms known as algae are a diverse category that includes both simple unicellular organisms and complex multicellular forms. They are distinct from plants, animals, and fungi and are classified in the Protista kingdom. There are many different types of habitats in which algae occur, including freshwater, marine, and terrestrial ecosystems. They can produce their own food because they are photosynthetic, meaning that light energy is converted into chemical energy during the process of photosynthesis.
Algae play an important role in the decomposition of organic waste and nutrient cycling in the soil-compost cycle. By secreting enzymes that break down complicated compounds, algae in the compost pile can accelerate the decomposition of organic material.
They can also absorb and store nutrients that are important for plant growth, such as nitrogen, phosphate and potassium.
Attached is a list from the paper (Dmytryk and Chojnacka 2018) for overview, I will not go into detail about each substance here.
Figure 24: The effect of poly- and oligosaccharides produced by algae when applied to plants. (Dmytryk and Chojnacka 2018)

Figure 25: The effect of phytohormones and hormone-like compounds when applied to plants. (Dmytryk and Chojnacka 2018)
Fungi (PGPFs)
The next huge component of soil microorganisms are fungi. Here, there is also a large number of different mechanisms of action. To cover the whole topic completely will again be very difficult in the short time available. Again, I recommend a review article:
A diverse group of fungi known as plant growth-promoting fungi (PGPFs) interact symbiotically with plants. They colonize roots and are critical for plant development, nutrient uptake, and tolerance to biotic and abiotic stresses. Many strains, such as the Glomeromycota, Ascomycota, and Basidiomycota, contain PGPFs.
Glomeromycota
The phylum Glomeromycota is known to play an important role in the development of arbuscular mycorrhizal (AM) associations with plants.
There are two types of mycorrhizal fungi, once endo and ectomycorrhiza. We need the endo variety as it forms symbiotic bonds with grasses and crops. The ecto strains are found on trees such as the common boletus in spruce. They settle on the surface of the roots and can form fruiting bodies.
In contrast, the arbuscular endo-mycorrhizal fungi we need are found in an intermediate layer of the root. Here they grow firmly and form a docking station, so to speak. From this, the fungus expands outward and forms tree-like looking hyphae (chains of fungal cells, like small roots). This is also the origin of the name of the arbuscular mycorrhizal fungi, because arbus comes from Latin and means tree.
Figure 26: Arbuscular arm of the fungus growing into the cell lumen. (Remy et al. 1994)
These fungi improve nutrient uptake and help plants cope with biotic and abiotic stress by colonizing the root cells of most plants. This increases the root surface area and thus promotes both water and phosphorus uptake considerably in combination with the release of organic acids and phosphatases. Phosphorus uptake radius was increased from several mm to as much as 11cm (JAKOBSEN et al. 1992). This is statistically significant and essential for optimal yield in the living soil system.
Rhizophagus irregularis, formerly known as Glomus intraradices, and the very prominent Glomus mossae, are examples of PGPF strains that promote growth of a variety of crops, including soybean, hemp, and tomato. (Błaszkowski 1994; Bever et al. 2001)
Ascomycota
Ascomycota is a phylum that includes a wide variety of fungi, many of which have been shown to have PGP properties. An example of a PGPF from this phylum is Trichoderma spp. which can promote plant growth by producing enzymes that degrade complex organic molecules and release growth-promoting substances such as indoleacetic acid (auxin hormone). Trichoderma can also improve plant tolerance to environmental stresses and strengthen plant defense mechanisms against pathogens (ISR -> see Chapter 2.3.2.1 Indirect Mechanisms).
Trichoderma is also known to produce a variety of enzymes, including cellulases, chitinases, proteases, and cellulases (CWDEs = cell wall degrading enzymes), which can hydrolyze the cell walls of other fungi and give them a competitive advantage over other fungal pathogens. These enzymes can support the degradation of organic material and release nutrients for plant uptake.
It is also important to know that different Trichoderma species have different plant infection capabilities. Trichoderma virens and Trichoderma koningii, for example, are known pathogens and can damage plants, while Trichoderma harzianum and Trichoderma asperellum promote plant growth.
Basidiomycota
Basidiomycota is another phylum that includes a variety of fungi, including mushrooms. An example of a PGPF from this phylum is Laccaria bicolor, which can promote plant growth by improving nutrient uptake and increasing plant tolerance to environmental stress. This fungus can also produce enzymes that degrade complex organic molecules, and has been shown to increase growth and root biomass in a number of plants.
Those interested in more facts and culture-specific characteristics of the strains should definitely read the review (Hossain et al. 2017) and (Devi et al. 2020) read.
Nematodes
Entomopathogenic nematodes (EPNs), commonly referred to as beneficial nematodes, are a variety of roundworms that live in the soil. They are known to contribute to soil improvement, inhibit pathogens and enhance plant development.
Beneficial nematodes come in a variety of forms, each with its own special characteristics and modes of action. The two genera most commonly used in biological control are Steinernema and Heterorhabditis. These nematodes are known to infest and kill a wide variety of invertebrates and soil-dwelling insects, including termites, pinworms and grubs. They infect their host through natural openings such as the mouth, anus, or respiratory pores. Once inside, they release symbiotic bacteria for them that destroy the host.
These nematodes are known to improve soil health by reducing insect populations and increasing nutrient availability.
Plant pathogenic nematodes can be suppressed by some beneficial nematodes, including species of the genus Phasmarhabditis. Known plant-parasitic nematodes such as root knotweed and dagger stick nematode can be infected and killed by these worms. When controlling plant-parasitic nematodes in soil, they can be used as biological control agents.
Because there are so many different beneficial uses, nematodes can be characterized by how they operate.

Figure 27: Systematics nematodes (Abd-Elgawad et al. 2017)
Again, as with the last chapters: Read reviews and form your own opinion (Abd-Elgawad et al. 2017) Chapters 1-3 should be suitable for this.
Furthermore, there is an exact listing of suitable hosts for the nematode species mentioned above. However, it is important to note, especially if you do not have a completely closed system, that you only use "native" species. Firstly for the sustainability mindset that one should have at least halfway and secondly due to the invasive nature of some species listed here.
Figure 28: Suitable hosts (insects) for Steinernema and Heterorhabditis sp. (Abd-Elgawad et al. 2017)
Protozoa
Protozoa are a diverse group of protozoa that play a critical role in the soil food web. They can be divided into several groups, including amoebae, ciliates and flagellates, each with different characteristics and ecological functions.
In the soil, protozoa consume bacteria, fungi and other microorganisms, regulating their populations. This consumption in turn affects the availability of nutrients such as nitrates and phosphates to plants. In addition, protozoa can also release nutrients from organic material, making them more available to plants. For example, Amoeba spp. and Vorticella spp. are known to play an important role in decomposing organic material and releasing nutrients.
But hormonal feedback is also sent to the plant. Auxin, for example, in the form of IAA, which is responsible for apical growth (growth along the top) of the roots, is released, suggesting to the plant that it can continue to expand in that direction, given protozoa, i.e., nutrient supply.
Figure 29: "A conceptual model illustrating the hormonal effects of microfauna on root growth, modified from Bonkowski & Brandt (2002). Root excretions (1) stimulate the growth of a diverse bacterial community (Bonkowski 2004)
Protozoa also play a role in controlling plant pathogens. Some species, such as Rhizomonas spp. consume bacteria that can cause plant diseases, helping to protect plants from infection.
One example of specific species that play a critical role in the soil food web is the genus Tetrahymena. These are ciliates that have the ability to break down cellulose in plant material to make it available for plant uptake and promote plant growth. This ability makes Tetrahymena a valuable biofertilizer for sustainable agriculture.
Protozoa fall into several categories based on their characteristics and ecological role. Below are some of the major groups of protozoa, examples of species within each group, and examples of how they have been shown to affect the soil food web and/or plant growth:
- Amoebae: Amoebae are single-celled organisms that use pseudopodia to move around and play an important role in decomposing organic matter and releasing nutrients. Examples of amoebae that have been shown to affect the soil food web and/or plant growth include Acanthamoeba spp. and Dictyostelium discoideum. Acanthamoeba spp. for example, have been shown to consume bacteria and fungi, regulating their populations; they also release nutrients from organic material, making them more available for plant uptake. Dictyostelium discoideum has been shown to be a soil protozoan that promotes plant growth by releasing phosphorus from organic material.
- Ciliates: Ciliates are unicellular organisms characterized by the presence of cilia, which they use for locomotion. Examples of ciliates that have been shown to affect the soil food web and/or plant growth are Tetrahymena spp. and Rhizomonas spp. Tetrahymena spp. has been shown to degrade cellulose in plant material, making it available for plant uptake and thus promoting plant growth; it also plays a role in the decomposition of organic matter and the release of nitrogen and phosphorus. Rhizomonas spp. have been shown to consume bacteria that can cause plant diseases, helping to protect plants from infection.
- Flagellates: Flagellates are unicellular organisms characterized by the presence of flagella, which they use for locomotion. Examples of flagellates that have been shown to affect the soil food web and/or plant growth are Vorticella spp. and Colpoda spp. Vorticella spp. has been found to consume bacteria, regulating their populations, and has also been found to release nitrogen and phosphorus into the soil, making them more available for plant uptake. Colpoda spp. has been shown to play a role in nutrient cycling by breaking down organic matter and releasing nutrients.
Worms
Earthworms, which belong to the phylum Annelida, play a critical role in the soil food web by improving soil structure, aerating soil, increasing its water-holding capacity, and recycling nutrients. They are classified into three main groups: epigean, endogean, and anoecious earthworms, each with unique characteristics and ecological functions. (Palm et al. 2013)
Earthworms are known to consume large amounts of soil and organic matter, thereby mixing and aerating the soil and increasing its porosity and water-holding capacity. This improves soil structure and fertility, which in turn benefits plant growth. However, this is again dependent on the soil itself. Worm digestion improved the new polyvalent bonds, but also loosens the clay bonds in the soil to some degree. That is, the higher the clay content of the soil, the less significant the improvement in soil structure. (Schrader and Zhang 1997) However, in our peat this is irrelevant due to the absence of clay and it can be said that worm humus generally improves structure.
One of the earthworms' most important contributions to nutrient cycling is their digestive system, a complex process that involves the release of enzymes and microbes. The earthworm gut contains several enzymes that break down organic material, including cellulase, which breaks down cellulose, and protease, which breaks down proteins. The earthworm gut also contains a diverse community of microbes, including bacteria and fungi that further break down organic matter.
The decomposition of organic material by earthworms leads to the release of various nutrients such as nitrogen, phosphorus and potassium, which are chelated in the earthworm gut. Chelated nutrients are minerals that are bound to organic molecules so that they can be better absorbed by plants. This process occurs through the action of enzymes called chelating agents, which are produced by the earthworms or their gut microbes. These chelating agents include siderophores and phytosiderophores, which chelate iron, and organic acids such as citrate, gluconate and oxalate, which chelate other metals such as zinc and manganese.
Epigeic earthworms such as Eisenia fetida (redworm) live on the surface of the soil and mainly decompose organic matter. They are often used in composting systems, where they efficiently break down organic waste and improve soil fertility, which benefits plant growth.
Endogeic earthworms, such as Aporrectodea spp, are burrowing organisms that live in the topsoil and subsoil. They create horizontal tunnels and improve soil structure by mixing organic matter and mineral soil, which promotes water infiltration and aeration. They also contribute to nutrient cycling by breaking down organic material and releasing nutrients, especially phosphorus and nitrogen, which are important for plant growth.
Anecious earthworms such as Lumbricus terrestris create deep vertical burrows in the soil that improve soil structure by increasing porosity and water infiltration, and they also bring deeper nutrients closer to the surface. Their burrows also serve as drainage channels and improve soil oxygenation.
In indoor soil systems, epigeic and endogenous earthworms can be useful for breaking down organic matter and improving soil fertility, while anecic earthworms can improve soil structure and aeration. By using a combination of earthworm species, indoor soil systems can benefit from the diverse ecological niches and functions of different earthworm groups, which in turn can lead to better plant growth.
Figure 30: Involvement of worms in pedogenesis (soil formation). (Babu Ojha and Devkota 2014)
It should be noted that earthworm populations and diversity may vary depending on the indoor system and management practices used. It is also important to have optimal moisture, temperature, and light conditions for the earthworm to survive and thrive. Also, consider the effects of applying pesticides or other chemicals that could harm the earthworm population.
In addition to their positive effects on soil structure and fertility, earthworms can also play an important role in regulating soil microbial communities. Earthworms have been shown to selectively feed on certain types of microorganisms and promote the growth of beneficial bacteria that can improve soil health and plant growth.
More detailed information can be found on google scholar or specifically in the review of (Lavelle 1988) or (Edwards and Arancon 2004)
Arthropods (arthropods)
Arthropods are a diverse group of animals that include insects, spiders, crustaceans, and millipedes. They play an important role in the soil food web, serving as decomposers, predators, and prey. They have various ecological functions and can have significant impacts on soil health and plant growth, especially in indoor living soil systems.
Arthropods important to the Soil food web include several groups of insects, such as beetles, springtails, and mites. Within these groups, there are many different species that can have different effects on soil health and plant growth.
For example, beetles of the Scarabaeidae family, such as the common dung beetle (Scarabaeus sp.), play an important role in decomposing animal waste and contribute to nutrient cycling in the soil. These beetles burrow into manure and help bury and decompose it, improving soil structure and fertility. However, we do not want them in our indoor soil, as we also do not use uncomposted/already turned animal products. This would in fact provide a perfect breeding ground for Salmonella ssp. and other harmful organisms that do not harm the plant, but are toxic to humans. This would even lead to flunking the purity tests of the flowers. (Deutscher Apotheker-Verlag Doktor Roland Schmiedel 2021; Kim and Jiang 2010).
Another group, the springtails (Collembola), are tiny, wingless insects that are common in soil. They are efficient decomposers and predators of other soil organisms and can help control pests and diseases in the soil. For specific information on this species, I recommend the website, which was really built with enthusiasm and contains really nice pictures, like this one (A Chaos of Delight 2023).
Figure 31: White springtail (A Chaos of Delight 2023)
Mites, especially from the Oribatida family, are common in soil and play an important role in breaking down organic matter and controlling pest populations. They feed on dead plant material and microorganisms, degrade them, release nutrients and contribute to the build-up of organic matter, which can have a positive effect on plant growth and soil health.
Due to our indoor soil and changing conditions from warm/humid to more cool/dry throughout the life cycle of the plant, climate specific arthropod populations change.
There are several arthropod groups and species that specifically thrive under hot and humid conditions, including those found in peaty soils. For example, some springtail species (Collembola) are known to tolerate high temperatures and humidity and are found in peat bogs and other wetlands. Certain mite species (Oribatida) are also common in peat soils and can play an important role in the decomposition of organic matter.
Other arthropods found in tropical, moist environments include weevils (Staphylinidae), which can play a role in the soil food web as decomposers, predators, and prey, and are found in a variety of habitats, including peat soils.
Thus, these groups thrive better in vegetative, warm/humid, conditions.
On the other hand, certain beetle species such as ground beetles (Carabidae) can tolerate low humidity in colder and drier conditions and are found in arid and semi-arid regions. As predators, they play an important role in the soil food web and can also be useful in pest control.
In addition, certain spider species, such as wolf spiders (Lycosidae), can tolerate low humidity and are found in a variety of habitats, including dry desert regions.
These would be species that we can use towards the end of flowering. Since they cope with it very well than resistant. However, there are also species that work but cannot perform their optimal performance under these conditions. But you can still use or even prefer them to the optimal performers if they have specific prey schemes, are easier to handle or have less impact on the bloom.
Phytoseiulus persimilis, a spider mite predator that tolerates low humidity and cool temperatures. This species is known as an effective biological control agent against spider mites, which are widespread in many crops and can thrive in dry environments.
Another example is Amblyseius californicus, a predatory mite that is effective in low humidity and low temperatures. It is known to feed on various mite and thrips species and has proven effective in greenhouses, nurseries and outdoors.
A third example is Neoseiulus cucumeris, a predatory mite species that feeds on thrips and spider mites. It tolerates both low humidity and low temperatures and is often used as a biological control agent for thrips in greenhouses and outdoors.
It is important to note that many arthropod populations are habitat specific, meaning that certain species thrive in certain soils and habitats. It would be beneficial to study the local arthropod populations in the target habitat before introducing a species, as it may not be able to survive or adapt to the new conditions.
In addition, a distinction can be made between microarthropods and macroarthropods, these differ mainly in their influence on the overall soil structure. Macroarthropods are capable of restructuring soil profiles or displacing large amounts of soil, while microarthropods usually inhabit the pore space in the soil (and play little part in its modification). Ants, in particular, are popular macroarthropods because their burrows almost count them as ecosystem engineers. They are of immense importance outdoors but again in the indoor system too aggressive towards the seedlings at the beginning and in their way to keep aphids as farm animals.(Neher & Barbercheck, 2019) Species which are really useful for us are e.g. spiders like the wolf spider, which is used e.g. also in cranberries as pathogen hunter. Here, however, care should be taken either to choose spiders that occur in our area or to keep the system so closed that an escape is not possible.
Ridding the soil of heavy metals by microbes
With a combination of different microbes, impurities in the soil such as heavy metals can be bound and removed. Heavy metals lead to failing health tests in commercial cultivation and cause severe nerve damage when consumed. This must be prevented and therefore a combined approach is needed to bind the heavy metals. The most important representatives here are:
- Cadmium
- Chrome
- Mercury
- Nickel
- Lead
- Copper
- Zinc
- Arsenic
Cadmium is allowed here in the lowest amount and zinc in the highest, but they should all be avoided preventively or bound curatively for the soil in microbes. Most heavy metals are also toxic to the species mentioned here, but these have evolved sophisticated mechanisms for detoxification by polymerization in cell walls or degradation by enzymes. This also makes them inaccessible to plants such as our hemp, which is particularly high in heavy metals. Most can even use these degradation products for their growth afterwards. Thus Pleurotus ostreatus decomposes crude oil among other things into potassium. The most important organisms for this are:
- Trichoderma ssp. = lead, cadmium, and arsenic. (Govarthanan et al. 2019; Zhang et al. 2018).
- Bacillus and Pseudomonas aeruginosa = zinc and copper. (Rajendran et al. 2003)
- Zooglea spp. and Citrobacter spp. = copper, nickel, cadmium, cobalt.(Verma and Kuila 2019)
- Citrobacter spp. and Chlorella vulgaris = cadmium, lead, uranium, copper, nickel, mercury, uranium. (Rajendran et al. 2003)
- Aspergilus Niger = cadmium, thorium, uranium, zinc. (Rajendran et al. 2003)
- Pleurotus ostreatus = cadmium, copper and zinc. (Rajendran et al. 2003)
- Rhizopus arrhizus = cadmium, mercury, lead.(Rajendran et al. 2003)
- Stereum hirsutum = cadmium, copper, cobalt, nickel (Verma and Kuila 2019)
- Phormidium valderium = cadmium; lead (Verma and Kuila 2019)
- Ganoderma applantus = copper, lead, mercury. (Verma and Kuila 2019)
The best review I have found on the general topic of bioremediation by white rot fungi is: (Rajendran et al. 2003). There are also many other uses listed, such as conversion of crude oil / motor oil to plant nutrients and environmentally safe minerals.
The most important bioremediation processes are bioaccumulation, bioleaching, biosorption, biotransformation, and biomineralization, as well as metal-microbe interactions. These processes allow heavy metals to be rendered harmless (Verma and Kuila 2019). However, for this to happen, some species that store it in their fruiting bodies must have it removed, otherwise, if the fungus dies, the metals will return to the soil.
Microorganisms are useful not only for metal dissolution, but also for the reduction and oxidation of transition metals.
Figure 32: Mechanisms of metal tolerance in microbes. (Rajendran et al. 2003)
Pathogenic pests
This chapter will probably be the one that we will consider the shortest, compared to its size. Because there is own training to the pest control professional and to summarize it would be really utopian.
We therefore limit ourselves to the organisms with the highest damage potentials and their spread, prevention and "cure". We look at 3 examples each for the 3 groups bacteria, viruses/viroids and fungi. But for this we need short definitions of the most important terms in plant protection.
Bacteria
Xanthomonas campestris pv. cannabis
Bacterial leaf spot disease of hemp is characterized by necrotic lesions about 1-2 mm in diameter with a yellow halo 2-3 mm wide on the leaves. It invades the plant through wounds (Netsu et al. 2014). In some crops such as tomato, it also induces black rot of stems, but I have not yet found a source on this effect specifically in cannabis.
Transmission occurs through contaminated seed, water, and equipment. Preventive measures include using disease-free seed, sterilizing equipment and avoiding overhead irrigation.
While the bacteria do not penetrate the seed, they can colonize the seed surface and persist there until germination. There they infect the seedling at the base and destroy it.
Curative measures include the use of chemical control methods such as copper hydroxide, streptomycin or oxytetracycline. However, this is not recommended in the Living soil system, as any life will also be destroyed. However, for mother plants of essential genetics that have no backup, sometimes you simply must resort to such means to achieve salvage. It is best to isolate the plant and spray only with the aid of adequate personal protective equipment (PPE) and approved quantity (see Fertilizer/Plant Protection Regulation).
Figure 33; Symptoms of Xanthomonas campestris pv. cannabis (Netsu et al. 2014)
Pseudomonas cannabina
This bacterium causes leaf spot disease and stem rot in cannabis plants. It infects the leaves, causing them to turn yellow and brown. Transmission occurs through contaminated water and equipment and is host specific. (Bull et al. 2010)
Preventive measures include the use of disease-free seed and sterilization of equipment. Curative measures include the use of chemical control methods such as copper hydroxide, streptomycin or oxytetracycline.
Phytoplasma (PH)
This group of bacteria causes witches' broom disease in cannabis plants. It infects the buds of the plant and causes them to stunt and form small, deformed flowers. Transmission usually occurs through insect vectors.
Preventive measures include controlling vector populations, using disease-free seed, and removing and destroying infected plant material.
Curative measures include the use of chemical control methods such as tetracycline or streptomycin (Chaube et al. 2015).
Viruses/Viroids
Latent hop viroid (HLVd)
This virus belongs to the potyvirus genus and causes latent infections in cannabis plants. Compared to viruses, viroids do not have a protective protein coat and are present as naked RNA strands.
This means the viroid can be present in the plant's virome but show no active symptoms until an appropriate stress or epigenetic changes cause the infection to break out. Symptoms include stunting, leaf malformation or chlorosis, brittle stems, and yield reduction up to 50%.
Chlorosis and yield reduction are among the non-specific symptoms that occur with many viruses/diseases. I.e. often the viroid cannot be identified directly by observation. For this, Rt-qPCR analysis for viroid RNA particles is necessary to really know that it is this specific viroid.
It is transmitted by contaminated seeds, pollen, vectors (insects that ingest it and transmit it when feeding on plants) and mechanically (scissors, hands, etc).
Preventive measures include absolute cleanliness of equipment, reduction of the plant-feeding insect population, and quarantine/testing when new cultivars are added to the plant. (Warren et al. 2019; Flores et al. 2005; Chiginsky et al. 2021)
So far, only meristem culture in combination with multiple testing on multiple plant organs and at different stages of the plant's life has promised a cure.
Through molecular breeding using molecular markers, high throughput sequencing technologies and large selection/diversity populations, resistant cultivars can be found and bred into elite lines. This requires deep expertise and large amounts of investment, but it would be feasible and would pay off its investment hundreds of times over. My master thesis will deal with this very topic and the first research work is already in progress.
A new emerging startup in this field is the In Vitro / Biotechnology company AlpineBioLabs, which is currently being founded. This will cover all areas of virus/viroid prevention, treatment and research on this topic.
After infection of a living soil bed with HLVd it is unfortunately necessary to remove the soil. There are very few studies on this, but it can be said that HLVd can only survive permanently in living plant parts. However, one often works with companion plants, which can then also be infected and thus provide shelter for the viroid. More detailed studies are needed to determine how long the viroid can survive in dead roots. Therefore, it is essential to prevent infection in the first place, the basis is a mature prevention concept in connection with clean starting material. This requires either an own breeding with monthly tests and quarantine programs or a partner who provides fresh, clean mother plants every three months and keeps an extensive, secure gene bank with backups.
Again, advertising on our own behalf, AlpineBioLabs will face these challenges and try to become the trusted partner for virus/viroid prevention/treatment in the DACH/EU region.
Lettuce chlorosis virus (LCV)
This virus belongs to the genus Criniviruses and the family Closteroviridae
It causes interveinal chlorosis, brittleness, and occasionally necrosis in older leaves on cannabis plants. It is transmitted by the whitefly vector. General symptoms include yellowing and stunted growth of the plant.
There are no cures for viral infections, so preventive measures such as the use of disease-free seed and equipment and control of the vector, the whitefly, are critical to controlling the disease.
As with almost all viruses/viroids, a meristem culture with integrated test protocol is the solution. Here, tissue that is not connected to the guidance channels (xylem/phloem) can be excised. This tissue (meristem) acts like stem cells and from them, any plant organ can be differentiated using organogenesis.
Figure 34: "Cannabis salatchlorosis virus (LCV) transmitted via shoots.(a) Stunted growth of a cannabis plant propagated from LCV-infected shoots (left) compared to growth of a cannabis plant propagated from uninfected shoots(right). (Hadad et al. 2019)
Cannabis cryptic virus (CanCV)
Cannabis Cryptic Virus (CanCV) is a virus that causes the symptoms of "hemp streak disease" in cannabis plants. The virus, which is currently the only known virus of the genus Caulimovirus infecting cannabis, was first identified in Italy in 2015.
Symptoms of CanCV infection include yellow or brown streaks on leaves, stunted growth and yield reduction. The virus is transmitted through the use of infected planting material and through contact with infected plant parts.
One of the most important effects of CanCV on horticulture is that it can significantly affect the yield and quality of cannabis plants. The virus can cause stunted growth, resulting in smaller plants with fewer flowers and lower THC and CBD levels.
Another impact of CanCV is that it can be difficult to control. So far, no studies on mechanical (horizontal) transmission are available. That is, so far it is thought to be transmitted exclusively vertically, i.e., via gametes (pollen/flowers) and thus seeds (Righetti et al. 2018)
Currently, there are no known chemical treatments for CanCV, and the most effective control measures involve the use of sanitation and virus-free planting material.
Again, meristem culture is the only curative solution. (Ziegler et al. 2012)
Fungi
Botrytis cinerea (BC)
This fungus causes gray mold on cannabis plants. It attacks the flowers, leaves and stems of the plant and causes wilting and death. It is transmitted by spores that are spread by wind or water. Preventive measures include using disease-free seeds, avoiding overhead irrigation, and removing and destroying infected plant material.
This fungus can infect both the live plant and drying flowers. Most will have already made acquaintance with it. The most important thing to avoid infection, besides preventing transmission, are the environmental conditions. In particular, free water, which occurs through condensation, provides the best breeding ground for Botrytis. It cannot survive on humidity alone. I.e. either plant sap has leaked out due to damage to the tissue (acute Ca2+ deficiency + mechanical action) or condensation is present.
The complete loss of medical cannabis in Israel due to B. cinerea is estimated to be 10% of total production in a year (Jerushalmi et al. 2020)
Alternaria alternata
Alternaria alternata is a fungal pathogen that can affect a variety of plant species, including cannabis. The fungus causes a disease called Alternaria leaf spot disease, which is characterized by the development of dark brown or black lesions on the leaves, stems and flowers of the plant.
The fungus infects the plant through natural openings such as stomata or wounds, but it can also enter through the seeds. Once the fungus has infected the plant, it begins to produce spores that are spread by wind and water, allowing the fungus to infect new plants.
Symptoms of Alternaria leaf spot disease include the formation of dark brown or black lesions on the leaves, stems, and flowers of the plant. These lesions can vary in size and shape and result in leaf yellowing, defoliation and death of the plant. The fungus can also infect buds and flowers, dramatically reducing yield and crop quality.
"In August 2000, for example, an outbreak of leaf spot disease in broadleaf tobacco (Nicotiana tabacum L.) caused by A. alternata resulted in the loss of 75% and 89% of total acreage in Connecticut and Massachusetts, respectively." (LaMondia 2001)
The fungus can survive on plant debris and survive in the soil for long periods of time. It is known that the fungus can infect plants at high relative humidity, so the disease is more pronounced in humid environments.
Figure 34: (A) Alternaria alternata (isolate 22A); (B) Botrytis cinerea (isolate 63A); and (C) control. (Jerushalmi et al. 2020)
Fusarium oxysporum/solani
Fusarium oxysporum and Fusarium solani are two different species of the fungus Fusarium known to cause plant diseases. While both species can infect a variety of plant hosts and cause similar symptoms e.g. cannabis, there are some differences in the symptoms they cause.
Fusarium oxysporum is known to cause a disease called Fusarium wilt, which is characterized by wilting and yellowing of the leaves and subsequent death of the plant. This pathogen infects the plant through the root system and colonizes the stem, causing wilting and death of the plant. Symptoms may also include leaf chlorosis (yellowing) and reduction in size of leaves, flowers and fruits.
Fusarium solani, on the other hand, is known to cause a disease called Fusarium crown rot, which is characterized by wilting and yellowing of the leaves and subsequent death of the plant. This pathogen infects the plant through the root system and colonizes the stem, causing wilting and death of the plant. Symptoms may also include leaf chlorosis, leaf necrosis (browning), and a reduction in the size of leaves, flowers, and fruits. To control these pathogens, it is important to use effective disease control strategies. These include regular monitoring of plants for symptoms of fungal infection, implementation of preventive measures such as crop rotation, sanitation, and the use of disease-free planting material, and the use of fungicides under the guidance of a plant pathologist or other qualified professional in extreme emergencies. (Ali Merjan 2017)
It is important to note that further research is needed to identify new and effective strategies to control these and other fungal pathogens that infect medical cannabis. In addition, research into the genetic and molecular characteristics of these pathogens could help identify new ways to control them. (Jerushalmi et al. 2020)
Figure 35: (A) Nutrient saline images of intact plants, including roots of (C) a plant inoculated with nutrient saline (control) compared to a plant inoculated with Fusarium oxysporum (isolate 64B) (D). (Jerushalmi et al. 2020)
These were now only pathogens (except botrytis) that affect living plants. But there are hundreds of others like Asperagillus ssp. etc. that can contaminate flowers and also have to be tested for according to the DAB.
Endophytes
"Understanding Cultivar Specificity and Soil Determinants of the Cannabis Microbiome." (Winston et al. 2014) and "Cannabis Microbiome and the Role of Endophytes in Modulating the Production of Secondary Metabolites: An Overview" (Taghinasab and Jabaji 2020) are two recent studies that examine the relationship between the endophytic microbiome of cannabis plants and their growth, as well as the effects of endophytes on the production of secondary metabolites in cannabis. Endophytes are microorganisms that live in the tissues of plants and can have various beneficial effects on their host, such as increased resistance to pathogens, enhanced nutrient uptake, and modulation of secondary metabolite production.
The first study, titled "Understanding Cultivar-Specificity and Soil Determinants of the Cannabis Microbiome," focused on two main factors that might influence the endophytic microbiome of cannabis plants: Cultivar Specificity and Soil Determinants. The researchers collected samples of cannabis plants grown in different soils and cultivars and used DNA sequencing techniques to identify and quantify the different endophytic microorganisms present.
The results showed that the endophytic microbiome of cannabis plants was indeed influenced by both cultivar specificity and soil factors. Different cannabis cultivars had different endophytic microbiomes, with some cultivars harboring a greater diversity of microorganisms than others. In addition, the endophytic microbiomes of cannabis plants grown in different soils were also different, with some soils supporting a greater diversity of endophytes than others. In particular, soils with higher organic matter content supported greater diversity of endophytic microorganisms, especially Proteobacteria, Actinobacteria, and Bacteroidetes. These endophytes have been shown to promote plant growth by producing plant growth-promoting compounds such as cytokinins and indoleacetic acid. In addition, soils with neutral pH (around 7) have been found to have higher levels of endophytes belonging to Phyla Firmicutes and Acidobacteria, which have been shown to enhance plant nutrient uptake.
The second study, "Cannabis Microbiome and the Role of Endophytes in Modulating the Production of Secondary Metabolites: A Review," focuses on the effects of endophytes on secondary metabolite production in cannabis. The study found that endophytic microorganisms can modulate the production of secondary metabolites in cannabis by affecting the expression of genes involved in the biosynthesis of these compounds. Specifically, endophytes belonging to the genus Burkholderia were found to positively correlate with the production of secondary metabolites in cannabis such as terpenoids and flavonoids. These endophytes have been shown to produce compounds such as siderophores and enzymes that can enhance the uptake of iron and other essential nutrients, which in turn could lead to an increase in the production of secondary metabolites.
Another hypothesis is the influence of hormone production by endophytes. The gibberellin produced positively influences the synthesis of THC/CBD (terpenoid-based substances) by regulating 1-aminocyclopropane-1-carboxylic acid levels, leading to an increase in ethylene levels. (Mansouri et al. 2011)
Figure 36: "The most common endophytes found in different tissues of Cannabis sativa plants from different geographic regions." (Robinson et al. 2016)
Outlook
Now this material should be enough to keep you busy for a few weeks, it took me several years of steady learning. You don't have to know everything or understand everything, but the better you understand your tools as a grower, the better your results will be.
In the next part, we will look at how to put this theoretical knowledge into practice in order to create your own soil mix. It will be about a deep analysis of the different substrates, aeration materials, compost forms, ferments, additives like Biochar and many more amendments. It is also filled with reference values to calculate the subsequent soil properties.
The final part will take place during the summer after the first field tests with the soil mix. Here the running conditions will be simulated and compared with soil tests. A test overview and calculation examples with correct field values will also be covered here, in order to finally arrive at a final recipe, which you can then reproduce yourself.
Bibliography
A Chaos of Delight (2023): All about Collembola/springtails- Neelipleona - A Chaos of Delight. Available online at https://www.chaosofdelight.org/all-about-collembola-neelipleona, last updated 11/01/2023, last checked 11/01/2023.
Abd-Elgawad, Mahfouz M. M.; Askary, Tarique Hassan; Coupland, James (2017): Biocontrol agents. Entomopathogenic and slug parasitic nematodes. Wallingford, Oxfordshire, UK, Boston, MA: CABI.
Agrios, George N. (2005): Plant pathology. 5th ed. Amsterdam: Elsevier Academic Press. Available online at http://www.sciencedirect.com/science/book/9780120445653.
ALBRECHT, WILLIAM A. (1959): Soil fertility and animal health. In: Soil Science 87 (5), p. 303. DOI: 10.1097/00010694-195905000-00014.
Ali Merjan (2017): The Fusarium Laboratory Manual. Available online at https://www.researchgate.net/publication/321385629_The_Fusarium_Laboratory_Manual.
Andrews, Richard (2013): cationexchange. Available online at http://www.zeoponix.com/cationexchange.htm, last updated 11/28/2013, last checked 09/01/2023.
Babu Ojha, Roshan; Devkota, Deepa (2014): Earthworms: 'Soil and Ecosystem Engineers' - a Review. In: WJAR 2 (6), pp. 257-260. DOI: 10.12691/wjar-2-6-1.
Becker, Florian (2019): Herzberg's two-factor theory of motivation. In: Motivating employees effectively: Springer, Berlin, Heidelberg, pp. 57-65. Available online at https://link.springer.com/chapter/10.1007/978-3-662-57838-4_8.
Bever, James D.; Schultz, Peggy A.; Pringle, Anne; Morton, Joseph B. (2001): Arbuscular Mycorrhizal Fungi: More Diverse than Meets the Eye, and the Ecological Tale of Why. In: BioScience 51 (11), p. 923. DOI: 10.1641/0006-3568(2001)051[0923:AMFMDT]2.0.CO;2.
Błaszkowski, J. (1994): Arbuscular fungi and mycorrhizae (Glomales) of the Hel Peninsula, Poland. In: Mycorrhiza 5 (1), pp. 71-88. DOI: 10.1007/BF00204022.
Blume, Hans-Peter; Scheffer, Fritz; Schachtschabel, Paul (eds.) (2010): Textbook of soil science. 16th, completely revised, updated and restructured ed. Heidelberg: Spektrum Akad. Verl. (Spektrum Textbook).
Bonkowski, Michael (2004): Protozoa and plant growth: the microbial loop in soil revisited. In: New Phytologist 162 (3), pp. 617-631. DOI: 10.1111/j.1469-8137.2004.01066.x.
Bull, Carolee T.; Manceau, Charles; Lydon, John; Kong, Hyesuk; Vinatzer, Boris A.; Fischer-Le Saux, Marion (2010): Pseudomonas cannabina pv. cannabina pv. nov., and Pseudomonas cannabina pv. alisalensis (Cintas Koike and Bull, 2000) comb. nov., are members of the emended species Pseudomonas cannabina (ex Sutic & Dowson 1959) Gardan, Shafik, Belouin, Brosch, Grimont & Grimont 1999. In: Systematic and Applied Microbiology 33 (3), pp. 105-115. DOI: 10.1016/j.syapm.2010.02.001.
Burgel, Lisa; Hartung, Jens; Graeff-Hönninger, Simone (2020): Impact of Different Growing Substrates on Growth, Yield and Cannabinoid Content of Two Cannabis sativa L. Genotypes in a Pot Culture. In: Horticulturae 6 (4), p. 62. DOI: 10.3390/horticulturae6040062.
Campbell, Sean M.; Anderson, Steven L.; Brym, Zachary T.; Pearson, Brian J. (2021): Evaluation of substrate composition and exogenous hormone application on vegetative propagule rooting success of essential oil hemp (Cannabis sativa L.). In: PLoS ONE 16 (7), e0249160. DOI: 10.1371/journal.pone.0249160.
Carol, Rachel J.; Dolan, Liam (2002): Building a hair: tip growth in Arabidopsis thaliana root hairs. In: Philosophical transactions of the Royal Society of London. Series B, Biological sciences 357 (1422), pp. 815-821. DOI: 10.1098/rstb.2002.1092.
Chaube, Savita; Kumar, Shailender; Dubey, Durgesh; Tiwari, Ajay Kumar; Upadhyaya, Parmatma Prasad; Rao, Govind Pratap (2015): Identification of a novel phytoplasma (16Sr XIV-A subgroup) associated with little leaf and witches' broom of Cannabis sativa L. ssp. sativa and C. sativa L. ssp. indica in India. In: Phytoparasitica 43 (2), pp. 275-279. DOI: 10.1007/s12600-014-0438-x.
Chet, I.; Ordentlich, A.; Shapira, R.; Oppenheim, A. (1990): Mechanisms of biocontrol of soil-borne plant pathogens by Rhizobacteria. In: Plant Soil 129 (1), pp. 85-92. DOI: 10.1007/BF00011694.
Chiginsky, Judith; Langemeier, Kaitlyn; MacWilliams, Jacob; Albrecht, Tessa; Cranshaw, Whitney; Fulladolsa, Ana Cristina et al. (2021): First Insights Into the Virus and Viroid Communities in Hemp (Cannabis sativa). In: Front. Agron. 3, article 778433, p. 96. DOI: 10.3389/fagro.2021.778433.
Cockson, Paul; Landis, Hunter; Smith, Turner; Hicks, Kristin; Whipker, Brian E. (2019a): Characterization of Nutrient Disorders of Cannabis sativa. In: Applied Sciences 9 (20), pp. 4432. DOI: 10.3390/app9204432.
Cockson, Paul; Landis, Hunter; Smith, Turner; Hicks, Kristin; Whipker, Brian E. (2019b): Characterization of Nutrient Disorders of Cannabis sativa. In: Applied Sciences 9 (20), pp. 4432. DOI: 10.3390/app9204432.
Couto, Daniel; Zipfel, Cyril (2016): Regulation of pattern recognition receptor signaling in plants. In: Nat Rev Immunol 16 (9), pp. 537-552. DOI: 10.1038/nri.2016.77.
Deutscher Apotheker Verlag (2021): German Pharmacopoeia. Official edition. Stuttgart, Eschborn, Berlin: Deutscher Apotheker Verlag; Avoxa - Mediengruppe Deutscher Apotheker; Akademie-Verl.
Deutscher Apotheker-Verlag Doctor Roland Schmiedel (2021): Deutsches Arzneibuch 2021 Digital. Official edition (DAB 2021). 1st edition. Stuttgart: Deutscher Apotheker Verlag.
Devi, Rubee; Kaur, Tanvir; Kour, Divjot; Rana, Kusam Lata; Yadav, Ashok; Yadav, Ajar Nath (2020): Beneficial fungal communities from different habitats and their roles in plant growth promotion and soil health. In: MBJ 5 (1), pp. 21-47. DOI: 10.21608/mb.2020.32802.1016.
Dmytryk, Agnieszka; Chojnacka, Katarzyna (2018): Algae As Fertilizers, Biostimulants, and Regulators of Plant Growth. In: Algae Biomass: Characteristics and Applications: Springer, Cham, pp. 115-122. Available online at https://link.springer.com/chapter/10.1007/978-3-319-74703-3_10.
Edwards, C. A.; Arancon, N. Q. (2004): Interactions among organic matter, earthworms, and microorganisms in promoting plant growth. In: Soil organic matter in sustainable agriculture, pp. 327-376.
Esteban, Raquel; Ariz, Idoia; Cruz, Cristina; Moran, Jose Fernando (2016): Review: Mechanisms of ammonium toxicity and the quest for tolerance. In: Plant science : an international journal of experimental plant biology 248, pp. 92-101. DOI: 10.1016/j.plantsci.2016.04.008.
Etesami, Hassan; Alikhani, Hossein Ali; Hosseini, Hossein Mirseyed (2015): Indole-3-acetic acid (IAA) production trait, a useful screening to select endophytic and rhizosphere competent bacteria for rice growth promoting agents. In: MethodsX 2, pp. 72-78. DOI: 10.1016/j.mex.2015.02.008.
Flores, Ricardo; Hernández, Carmen; Martínez de Alba, A. Emilio; Daròs, José-Antonio; Di Serio, Francesco (2005): Viroids and viroid-host interactions. In: Annual review of phytopathology 43, pp. 117-139. DOI: 10.1146/annurev.phyto.43.040204.140243.
Geelen, P. A. M.; Voogt, J. O.; van Weel, P. A. (2019): Plant empowerment. The basic principles : how an integrated approach based on physics and plant physiology leads to balanced growing methods for protected crops resulting in healthy resilient plants, high yield and quality, low energy costs and economic greenhouse concepts. [Vlaardingen]: Letsgrow.com.
Goswami, Dweipayan; Thakker, Janki N.; Dhandhukia, Pinakin C. (2016): Portraying mechanics of plant growth promoting rhizobacteria (PGPR): A review. In: Cogent Food & Agriculture 2 (1). DOI: 10.1080/23311932.2015.1127500.
Govarthanan, M.; Mythili, R.; Kamala-Kannan, S.; Selvankumar, T.; Srinivasan, P.; Kim, H. (2019): In vitro bio-mineralization of arsenic and lead from aqueous solution and soil by wood rot fungus, Trichoderma sp. In: Ecotoxicology and Environmental Safety 174, pp. 699-705. DOI: 10.1016/j.ecoenv.2019.03.034.
Hadad, Lior; Luria, Neta; Smith, Elisheva; Sela, Noa; Lachman, Oded; Dombrovsky, Aviv (2019): Lettuce chlorosis virus disease: a new threat to cannabis production. In: Viruses 11 (9), p. 802. DOI: 10.3390/v11090802.
Hammer, P. E.; Hill, D. S.; Lam, S. T.; van Pée, K. H.; Ligon, J. M. (1997): Four genes from Pseudomonas fluorescens that encode the biosynthesis of pyrrolnitrin. In: Applied and Environmental Microbiology 63 (6), pp. 2147-2154. DOI: 10.1128/aem.63.6.2147-2154.1997.
Hartemink, Alfred E.; Barrow, N. J. (2023): Soil pH - nutrient relationships: the diagram. In: Plant Soil, pp. 1-7. DOI: 10.1007/s11104-022-05861-z.
Helliwell, J. R.; Miller, A. J.; Whalley, W. R.; Mooney, S. J.; Sturrock, C. J. (2014): Quantifying the impact of microbes on soil structural development and behavior in wet soils. In: Soil Biology and Biochemistry 74, pp. 138-147. DOI: 10.1016/j.soilbio.2014.03.009.
Hossain, Md. Motaher; Sultana, Farjana; Islam, Shaikhul (2017): Plant growth-promoting fungi (PGPF): phytostimulation and induced systemic resistance. In: Plant-Microbe Interactions in Agro-Ecological Perspectives: Springer, Singapore, pp. 135-191. Available online at https://link.springer.com/chapter/10.1007/978-981-10-6593-4_6.
International Peatland Society (2019): What is peat? - International Peatland Society. Available online at https://peatlands.org/peat/peat/, last updated 02/07/2019, last checked 16/04/2023.
JAKOBSEN, I.; ABBOTT, L. K.; ROBSON, A. D. (1992): External hyphae of vesicular-arbuscular mycorrhizal fungi associated with Trifolium subterraneum L. 1. Spread of hyphae and phosphorus inflow into roots. In: New Phytologist 120 (3), pp. 371-380. DOI: 10.1111/j.1469-8137.1992.tb01077.x.
Jerushalmi, Shachar; Maymon, Marcel; Dombrovsky, Aviv; Freeman, Stanley (2020): Fungal Pathogens Affecting the Production and Quality of Medical Cannabis in Israel. In: Plants 9 (7), p. 882. DOI: 10.3390/plants9070882.
Kim, J.; Jiang, X. (2010): The growth potential of Escherichia coli O157:H7, Salmonella spp. and Listeria monocytogenes in dairy manure-based compost in a greenhouse setting under different seasons. In: Journal of Applied Microbiology 109 (6), pp. 2095-2104. DOI: 10.1111/j.1365-2672.2010.04841.x.
Küstermann, B., Kainz, M. & Hülsbergen, K.‑J. (2008). Modeling carbon cycles and estimation of greenhouse gas emissions from organic and conventional farming systems. Renewable Agriculture and Food Systems, 23(1), 38–52. https://doi.org/10.1017/S1742170507002062
LaMondia, J. A. (2001): Outbreak of Brown Spot of Tobacco Caused by Alternaria alternata in Connecticut and Massachusetts. In: Plant Disease 85 (2), p. 230. DOI: 10.1094/PDIS.2001.85.2.230B.
Landi, S. (1997): Mineral nutrition of Cannabis sativa L. In: Journal of Plant Nutrition 20 (2-3), pp. 311-326. DOI: 10.1080/01904169709365252.
Lavelle, P. (1988): Earthworm activities and the soil system. In: Biol Fert Soils 6 (3), pp. 237-251. DOI: 10.1007/BF00260820.
Li, Wei; Xiang, Fen; Zhong, Micai; Zhou, Lingyun; Liu, Hongyan; Li, Saijun; Wang, Xuewen (2017): Transcriptome and metabolite analysis identifies nitrogen utilization genes in tea plant (Camellia sinensis). In: Sci Rep 7 (1), p. 1693. DOI: 10.1038/s41598-017-01949-0.
Lin, Y. S.; Heuer, V. B.; Ferdelman, T. G.; Hinrichs, K.-U. (2010): Microbial conversion of inorganic carbon to dimethyl sulfide in anoxic lake sediment (Plußsee, Germany). In: Biogeosciences 7 (8), pp. 2433-2444. DOI: 10.5194/bg-7-2433-2010.
Llewellyn, David; Golem, Scott; Jones, A. Maxwell P.; Zheng, Youbin (2023): Foliar Symptomology, Nutrient Content, Yield, and Secondary Metabolite Variability of Cannabis Grown Hydroponically with Different Single-Element Nutrient Deficiencies. In: Plants 12 (3), p. 422. DOI: 10.3390/plants12030422.
LOACH, K. (1985): Rooting of cuttings in relation to the propagation medium. In: (35), pp. 472-485. Available online at https://pascal-francis.inist.fr/vibad/index.php?action=getRecordDetail&idt=8244846.
Local Land Services (NSW Government) (2020): Cation Exchange Capacity. Fact Sheet 4.
Logan Labs (2023): Logan Labs Soil Testing Services | Lakeview Ohio, last updated 04/01/2023, last verified 04/01/2023.
Lowenfels, Jeff; Lewis, Wayne (eds.) (2016): Teaming with microbes. The organic gardener's guide to the soil food web. Revised edition. Portland, Or.: Timber Press.
Maathuis, Frans J. M.; Diatloff, Eugene (2013): Roles and functions of plant mineral nutrients. In: Methods in molecular biology (Clifton, N.J.) 953, pp. 1-21. DOI: 10.1007/978-1-62703-152-3_1.
Mansouri, Hakimeh; Asrar, Zahra; Amarowicz, Ryszard (2011): The response of terpenoids to exogenous gibberellic acid in Cannabis sativa L. at vegetative stage. In: Acta Physiol Plant 33 (4), pp. 1085-1091. DOI: 10.1007/s11738-010-0636-1.
Naitam, Mayur G.; Kaushik, Rajeev (2021): Archaea: An Agro-Ecological Perspective. In: Current microbiology 78 (7), pp. 2510-2521. DOI: 10.1007/s00284-021-02537-2.
Natural Resources Conservation Service (2023): Soil Classification | Natural Resources Conservation Service. Available online at https://www.nrcs.usda.gov/resources/guides-and-instructions/soil-classification, last updated Jan. 11, 2023, last checked Jan. 11, 2023.
Netsu, Osamu; Kijima, Toshio; Takikawa, Yuichi (2014): Bacterial leaf spot of hemp caused by Xanthomonas campestris pv. cannabis in Japan. In: J Gen Plant Pathol 80 (2), pp. 164-168. DOI: 10.1007/s10327-013-0497-8.
Niu, Dong-Dong; Wang, Chun-Juan; Guo, Ya-Hui; Jiang, Chun-Hao; Zhang, Wen-Zhi; Wang, Yun-peng; Guo, Jian-Hua (2012): The plant growth-promoting rhizobacterium Bacillus cereus AR156 induces resistance in tomato with induction and priming of defense response. In: Biocontrol Science and Technology 22 (9), pp. 991-1004. DOI: 10.1080/09583157.2012.706595.
Oswald, Iain W. H.; Ojeda, Marcos A.; Pobanz, Ryan J.; Koby, Kevin A.; Buchanan, Anthony J.; Del Rosso, Josh et al. (2021): Identification of a New Family of Prenylated Volatile Sulfur Compounds in Cannabis Revealed by Comprehensive Two-Dimensional Gas Chromatography. In: ACS Omega 6 (47), pp. 31667-31676. DOI: 10.1021/acsomega.1c04196.
Palm, Juliane; van Schaik, N. Loes M.B.; Schröder, Boris (2013): Modeling distribution patterns of anecic, epigeic and endogeic earthworms at catchment-scale in agro-ecosystems. In: Pedobiologia 56 (1), pp. 23-31. DOI: 10.1016/j.pedobi.2012.08.007.
Pandey, Nalini (2018): Role of Plant Nutrients in Plant Growth and Physiology. In: Plant Nutrients and Abiotic Stress Tolerance: Springer, Singapore, pp. 51-93. Available online at https://link.springer.com/chapter/10.1007/978-981-10-9044-8_2.
Park, K-H; Lee, C-Y; Son, H-J (2009): Mechanism of insoluble phosphate solubilization by Pseudomonas fluorescens RAF15 isolated from ginseng rhizosphere and its plant growth-promoting activities. In: Letters in applied microbiology 49 (2), pp. 222-228. DOI: 10.1111/j.1472-765X.2009.02642.x.
Peterson, J. C. (1982): Effects of pH [hydrogen-ion concentration] upon nutrient availability in a commercial soilless root medium utilized for floral crop production. Available online at https://agris.fao.org/agris-search/search.do?recordid=us19830871517.
Płociniczak, T.; Kukla, M.; Wątroba, R.; Piotrowska-Seget, Z. (2013): The effect of soil bioaugmentation with strains of Pseudomonas on Cd, Zn and Cu uptake by Sinapis alba L. In: Chemosphere 91 (9), pp. 1332-1337. DOI: 10.1016/j.chemosphere.2013.03.008.
Rajendran, P.; Muthukrishnan, J.; Gunasekaran, P. (2003): Microbes in heavy metal remediation. In: 0975-1009. Available online at https://nopr.niscpr.res.in/handle/123456789/17153.
Rasool, Saiema; Ahmad, Altaf; Siddiqi, T. O.; Ahmad, Parvaiz (2013): Changes in growth, lipid peroxidation and some key antioxidant enzymes in chickpea genotypes under salt stress. In: Acta Physiol Plant 35 (4), pp. 1039-1050. DOI: 10.1007/s11738-012-1142-4.
Remy, W.; Taylor, T. N.; Hass, H.; Kerp, H. (1994): Four hundred-million-year-old vesicular arbuscular mycorrhizae. In: Proceedings of the National Academy of Sciences of the United States of America 91 (25), pp. 11841-11843. DOI: 10.1073/pnas.91.25.11841.
Righetti, Laura; Paris, Roberta; Ratti, Claudio; Calassanzio, Matteo; Onofri, Chiara; Calzolari, Davide et al. (2018): Not the one, but the only one: about Cannabis cryptic virus in plants showing 'hemp streak' disease symptoms. In: Eur J Plant Pathol 150 (3), pp. 575-588. DOI: 10.1007/s10658-017-1301-y.
Rillig, Matthias C.; Mummey, Daniel L. (2006): Mycorrhizas and soil structure. In: The New phytologist 171 (1), pp. 41-53. DOI: 10.1111/j.1469-8137.2006.01750.x.
Robinson, Rebekah J.; Fraaije, Bart A.; Clark, Ian M.; Jackson, Robert W.; Hirsch, Penny R.; Mauchline, Tim H. (2016): Wheat seed embryo excision enables the creation of axenic seedlings and Koch's postulates testing of putative bacterial endophytes. In: Sci Rep 6 (1), pp. 25581. DOI: 10.1038/srep25581.
Sasse, Joelle; Martinoia, Enrico; Northen, Trent (2018): Feed Your Friends: do plant exudates shape the root microbiome? In: Trends in Plant Science 23 (1), pp. 25-41. DOI: 10.1016/j.tplants.2017.09.003.
Schrader, Stefan; Zhang, Haiquan (1997): Earthworm casting: Stabilization or destabilization of soil structure? In: Soil Biology and Biochemistry 29 (3-4), pp. 469-475. DOI: 10.1016/S0038-0717(96)00103-4.
St. Croix Sensory Inc (2018): Parameters of Odor. In: St. Croix Sensory, Inc., 03.01.2018. Available online at https://www.fivesenses.com/Documents/Library/StCroixSensory_Odor_Parameters_Copyright2018.pdf, last checked 19.09.2022.
Stevenson, Frank J. (1994): Humus chemistry. Genesis, composition, reactions. 2nd ed. New York: Wiley.
Taghinasab, Meysam; Jabaji, Suha (2020): Cannabis Microbiome and the Role of Endophytes in Modulating the Production of Secondary Metabolites: An Overview. In: Microorganisms 8 (3), p. 355. DOI: 10.3390/microorganisms8030355.
Tegeder, Mechthild; Rentsch, Doris (2010): Uptake and partitioning of amino acids and peptides. In: Molecular plant 3 (6), pp. 997-1011. DOI: 10.1093/mp/ssq047.
Turunen, M.; Hyväluoma, J.; Heikkinen, J.; Keskinen, R.; Kaseva, J.; Koestel, J.; Rasa, K. (2019): Quantifying Physical Properties of Three Sphagnum -Based Growing Media as Affected by Drying-Wetting Cycles. In: Vadose zone j. 18 (1), pp. 190033. DOI: 10.2136/vzj2019.04.0033.
Verma, Samakshi; Kuila, Arindam (2019): Bioremediation of heavy metals by microbial process. In: Environmental Technology & Innovation 14, pp. 100369. DOI: 10.1016/j.eti.2019.100369.
Wang, H.; Inukai, Y.; Yamauchi, A. (2006): Root Development and Nutrient Uptake. In: Critical Reviews in Plant Sciences 25 (3), pp. 279-301. DOI: 10.1080/07352680600709917.
Warren, J. G.; Mercado, J.; Grace, D. (2019): Occurrence of Hop Latent Viroid Causing Disease in Cannabis sativa in California. In: Plant Disease 103 (10), p. 2699. DOI: 10.1094/PDIS-03-19-0530-PDN.
Weigend, Maximilian; Mustafa, Adeel; Ensikat, Hans-Jürgen (2018): Calcium phosphate in plant trichomes: the overlooked biomineral. In: Planta 247 (1), pp. 277-285. DOI: 10.1007/s00425-017-2826-1.
Winston, Max E.; Hampton-Marcell, Jarrad; Zarraonaindia, Iratxe; Owens, Sarah M.; Moreau, Corrie S.; Gilbert, Jack A. et al. (2014): Understanding cultivar-specificity and soil determinants of the cannabis microbiome. In: PLOS ONE 9 (6), e99641. DOI: 10.1371/journal.pone.0099641.
Zhang, Xu; Li, Xinxin; Yang, Huanhuan; Cui, Zhaojie (2018): Biochemical mechanism of phytoremediation process of lead and cadmium pollution with Mucor circinelloides and Trichoderma asperellum. In: Ecotoxicology and Environmental Safety 157, pp. 21-28. DOI: 10.1016/j.ecoenv.2018.03.047.
Ziegler, Angelika; Matoušek, Jaroslav; Steger, Gerhard; Schubert, Jörg (2012): Complete sequence of a cryptic virus from hemp (Cannabis sativa). In: Arch Virol 157 (2), pp. 383-385. DOI: 10.1007/s00705-011-1168-8.
FFJ - Fermented Fruit Juice - The terp booster from Korean Natural Farming
Why do I need Fermented Fruit Juice?
The name says it all: FFJ is the sieved juice from homofermentative converted fruits which can be produced with the help of fermentation without the formation of alcohols. Depending on the time of harvest, these contain different proportions of relevant nutrients. (Ju-young Cho 1992)
In particular, these are the molecules that have not yet been completely broken down into their basic building materials (NO3-), such as amino acids and polysaccharides. These can be absorbed directly by the plant and save it, so to speak, binding energy, since it no longer has to synthesize it itself.
The homofermentative destruction (see LAB article for a detailed explanation) is particularly important here, as we only want to receive lactate and acetate in the first steps of lactic acid fermentation, as alcohol would kill the responsible microbes.
Which fruit can I use?
The most important thing in the production of FFJ is to use locally homegrown or at least Demeter / organically grown fruits, whereby the latter will perform significantly worse. That's because for the planned fermentation we need a large number of microbes, especially Lactobacillus, which we get more by adding LAB. Natural yeasts and other microbes help in this process.
Once we have fulfilled this point, we can concentrate on the fruit. Nutrient analyzes can be used here, as these substances are then released into the liquid through micronization (decomposition of the plant tissue during fermentation).
High concentrations of potassium make apples, for example, a well-suited candidate for our FFJ. But it doesn't just have to be the obvious fruits; vegetables like the pumpkin also achieve excellent results. In principle, all kinds of fruit can be used as long as they contain one of the 17 elements that can be used by plants.
Figure 1: Composition of mineral content of different apple varieties (Henríquez et al. 2010)
Immature or overripe - which is better in which phase?
When choosing the fruit, special attention must be paid to the degree of ripeness. This determines the timestamp of use in the growth cycle of cannabis in flowering phase. The flowering phase can generally be divided into three phases.
The first is the formation of inflorescences, paired with temporary, rapidly increasing elongation growth. Here the plant needs the highest amount of phosphorus in the flowering phase in order to produce enough inflorescences. Unripe fruits are the best choice for our FFJ, as they contain a high amount of oxalic acid. This organic acid is even better suited for dissolving phosphorus than sulfuric acid.. (Mendes et al. 2020)
Figure 2: High oxalic acid content in unripe bananas (Heather Wyman und Palmer 1964)
Overripe, but not yet bad fruit is excellently suited for use as an addition to irrigation water from the main phase, also known as "bulk weeks", ie "rapid biomass growth". These have the highest levels of organic acids, especially malic and citric acid.
Figure 3: Organic acid content in ripe banana (Heather Wyman und Palmer 1964)
These acids have already shown in several studies to significantly increase the dry weight of plants in particular in a concentration of 100mg/L (Talebi et al. 2014)
If you look at Figure 1, you will find that ripe bananas contain, for example, approx. 600mg / ml (6.2meq / 100g) of magic acid. This means that even small concentrations of FFJ are sufficient in the application.
But that was not all: Malic acid also strengthens the symbiosis with benign rhizobacteria such as B.subtilis FB17, which increases endogenous pathogen tolerance. These acids are normally produced in a complex manner by the plant itself. This also saves plant energy as ATP, which can be used for other processes. (Rudrappa et al. 2008)
How is FFJ made?
We've already covered the main part, but you need a few things first before you can start. Here is the list of ingredients you need:
- The fruit (unripe or overripe)
- A large mason jar (preferably two liters)
- Cane sugar (for every gram of fruit, add one gram of sugar)
- A rubber band
- A cover, like a paper towel, that allows gas to be exchanged.
- A bowl to mix
- A scale
First cut the fruit into thumb-sized pieces and weigh them into the bowl. Now add the equivalent amount of sugar and a shot (2-4ml) of LAB.
Then mix it vigorously so that everything is moistened with sugar. You pour the mixture into the glass until it is too full. The edges / places with sugar on the outside of the glass can be cleaned with vinegar, otherwise ants or vermin could be interesting in the glass. You complete this with the paper towel and the rubber. Then it should be placed in a warm, dark place.
After 3-5 days, the solid mass should have lifted from the liquid. Simply sieve and saturate the liquid again with a little sugar.
How is FFJ used and applied?
Our FFJ is ready now. The leftovers can either be incorporated as head fertilization (fertilizer on substrate), but pay attention to the amount, otherwise you can catch unwanted guests.
You can also use the liquid normally in the maintenance spray (FPJ + brown rice vinegar + OHN), i.e. with rice vinegar and OHN. The concentration remains at 1: 500 as with FPJ. Read more about OHN in our first Korean Natural farming article.
Foliar fertilization should only be used if there are still small or non-existent blooms, as otherwise there is a risk of mold. As a rule of thumb, flowering week 2-3 is the last opportunity to apply FFJ to leaves.
Bibliography
Heather Wyman; Palmer, James K. (1964): Organic Acids in the Ripening Banana Fruit. In: Plant Physiology 39 (4), S. 630–633. Online verfügbar unter http://www.jstor.org/stable/4260273.
Henríquez, Carolina; Almonacid, Sergio; Chiffelle, Italo; Valenzuela, Tania; Araya, Manuel; Cabezas, Lorena et al. (2010): Determination of Antioxidant Capacity, Total Phenolic Content and Mineral Composition of Different Fruit Tissue of Five Apple Cultivars Grown in Chile. In: Chilean J. Agric. Res. 70 (4), S. 523–536. DOI: 10.4067/S0718-58392010000400001.
Ju-young Cho (1992): Cho`s Natural farming: Recipes and Instructions for use. Japan: Modern Agriculture.
Mendes, Gilberto de Oliveira; Murta, Hiunes Mansur; Valadares, Rafael Vasconcelos; Da Silveira, Wendel Batista; Da Silva, Ivo Ribeiro; Costa, Maurício Dutra (2020): Oxalic acid is more efficient than sulfuric acid for rock phosphate solubilization. In: Minerals Engineering 155, S. 106458. DOI: 10.1016/j.mineng.2020.106458.
Rudrappa, Thimmaraju; Czymmek, Kirk J.; Paré, Paul W.; Bais, Harsh P. (2008): Root-secreted malic acid recruits beneficial soil bacteria. In: Plant Physiol 148 (3), S. 1547–1556. DOI: 10.1104/pp.108.127613.
Talebi, Majid; Hadavi, Ebrahim; Jaafari, Nima (2014): Foliar Sprays of Citric Acid and Malic Acid Modify Growth, Flowering, and Root to Shoot Ratio of Gazania (Gazania rigens L.): A Comparative Analysis by ANOVA and Structural Equations Modeling. In: Advances in Agriculture 2014, S. 1–6. DOI: 10.1155/2014/147278.
FPJ (Fermented plant juice) - the KNF terp booster for Cannabis
1. What is FPJ and why do I need it?
Many people make little to no use of their green waste or weeds. The highest of feelings is the compost pile, which for most is a slow rotting compost. This requires long waiting periods. A thermophilic compost is faster, but also destroys important enzymes and amino acids through temperature exposure.
To preserve these essential plant nutrients, an enzymatic or fermentative conversion is excellent. This gentle, cold process is not only fast (less than two weeks) but also space-saving and odorless.
The amino acids obtained in this way, such as glutamine, can be used directly by the plant and do not first have to be synthesized from nitrate with an energetic (ATP) effort. This feeds the plant and provides it with energy-rich food.
1: Assimilation of amonia
Enzymes such as glucanases and amylases help to convert the contained secondary nutrients, like polysaccharides and amino acids, into usable substances.
Furthermore, a broad spectrum of probiotic microbes is present in the finished FPJ. These colonize the plant surface and the soil. In doing so, they displace pathogens and promote beneficial nutrient converters such as PSB (phosphorus solubilizing bacteria).
2. What plants are suitable for FPJ?
First we need to collect plants or plant in the garden for specific use. Care should be taken that the plant is very watery, otherwise complete fermentation will not occur.
Examples would be fresh, pathogen-free cuttings of tomatoes or dandelions.
Both are particularly suitable due to their nutrient accumulation in the tissue. Other plants are Beinweil, hemp leaves (only large, thick awnings) or flowers of heavy eating plants like zucchini, eggplant or bananas
If you want to make it more specific, you can just look online to see what nutrients are included. However, the water content and other categories must of course be respected there.
An important point with collected plants is the time of collection. Here you should go out early in the morning to be able to harvest the plants with morning dew. This will give you the greatest density/diversity of microbes. (Source 2)
3. How FPJ is made
The production is very simple and cheap. For 0.5L FPJ you need the following utensils:
- 1x 1L jar
- Approx. 500-800G brown unrefined sugar (can also be made with normal sugar)
- Note: Unrefined sugar still contains its molasses content
- Note: Approx. weight because you need the same amount of sugar as plant matter
- Enough plant matter to fill your ¾ glass jar.
- Mixing bowl
- 1-2 tablespoons of LAB serum (speeds up the process, but is not essential).
- Paper towel or a tissue that breathes
- Rubber band
- Dark storage space
Now that you have collected your material, you will need to chop it into thumb-sized pieces. It is best to shred the plant tissue directly in your bowl so that no juice is lost. This is essential for complete fermentation. Tearing provides a larger surface area for the sugar to attach to. Now we add the same amount of sugar as plant material and mix vigorously. It sucks the water out of the tissue and provides an excellent food source for our microbes. Then we add a shot of LAB, this stimulates the degradation processes by starting the homofermentative Lactic acid fermentation. Pour the resulting pulp into our jar until it is ¾ full and press it down evenly so that there is no air left in the mass. Make sure you really get everything out of the bowl, otherwise you will lose valuable juice.
On top of the pressed down parts of the plant, we add another 1-2cm layer of sugar to seal it and prevent the growth of unwanted fungi.
Tie the paper towel or a coffee filter (if the jar is small enough) with the rubber over the opening of the jar to allow gas exchange to continue. After all, CO2 is produced during fermentation. The rims must be clean, otherwise flies will be attracted. The best and most microbe-friendly cleaning you get with vinegar. This should be in the repertoire of every Korean Natural Farmer.
4. How is FPJ applied?
Like most Korean Natural Farming remedies, FPJ should be given in combination with others. A table on this can be found in the first part of the article series.
FPJ is mostly given as a "Maintenace Spray". This includes FPJ (1:500), OHN (1:1000) and BRV (1:500).
FPJ acts as the nutritional part here, OHN as pathogen prevention and terpene stimulant (Source 3). The vinegar contains many important amino acids and buffers the PH on the leaf surface. It thus ensures the rapid conversion of the remaining sugar on the leaf.
Therefore, BRV is indispensable in the administration of FPJ, otherwise pests will be attracted.
5. At what stage of my cannabis plant do I give FPJ?
Maintenace spray can be given from the end of the seedling phase until the second week of the flowering phase. The application is done by foliar fertilization. It is important not to use an atomizer but a normal pump sprayer, otherwise the microbes will burst due to the pressure and the small openings.
Spraying should not be done during full light or high temperature, otherwise there is no possibility of transpiration. This leads to overheating of the plant and corresponding reduction of photosynthetic performance.
It is also possible to apply with the help of watering, but not as efficient as via foliar fertilization. The concentrations remain the same.
6. The importance of local raw materials
Now we have gone through the topic FPJ, which can be meanly called Vegbooster. We will deal with the flower counterpart in the next article.
As a small conclusion, I would like to emphasize again the importance of local ingredients. The plant material must come from your area or at least your climate zone, otherwise the microbes are not optimally adapted. This will cause you to lose tremendous potential. Bought plants are also often contaminated with pesticides, which destroys the whole concept of KNFs.
7. Quellen
1. Spektrum.de; „Ammoniakassimilation“, (https://www.spektrum.de/lexikon/biochemie/ammoniakassimilation/338)
2. „Fermented Plant Juice for Cannabis“; (https://rollinggreenacres.com/fermented-plant-juice-for-cannabis/)
3. The_worlds_last_hope; (https://www.instagram.com/the_worlds_last_hope/)
LAB - lactic acid bacteria I The bouncer of Korean Natural Farming (KNF)
What are lactic acid bacteria actually?
The term lactic acid bacteria is the German translation for Lactic Acid Bacteria. This English term is used internationally in the Korean Natural Farming scene. Lactic acid bacteria can be isolated via fermentation processes and used for garden applications. We will go into more detail about the exact application possibilities in the next chapter, but first let's look at the structure and taxonomy of the microbes.
LAB are a large group of bacteria and cannot be narrowed down to a single species. They have been used for centuries by many different cultures for fermentation and preservation of foods such as sauerkraut or kimchi. These bacteria survive even the low/acidic PH produced by fermentation, which also allows them to survive our digestive system when consumed. These bacteria are very good for our intestines and help with constipation and keep the microorganism milieu in balance.
As if that wasn't enough, LABs are also responsible for the production of yogurt and cheese, as they form the basis for separating the rennet from the liquid. However, in today's high-tech production, native rennet strains are no longer used but specially grown cultures. This is because different population compositions also produce the different types of cheese.
What are LAB used for in Korean Natural Farming?
Now we have heard a lot about commercial use, but how does it benefit us now in gardening? Since there are three different purposes, we will make a small separate chapter for each.
Use as a single preparation
Now that we have isolated our LAB serum, we can use it as is. The only thing to keep in mind is to dilute it with water, otherwise it can give off a rather strong odor after application. The rate for this is about 1:1000 with pure, antibiotic-free water. Unfortunately, an exact figure cannot be given here, as each LAB serum is colonized to a different extent.
It can then be sprayed directly onto the soil around the base of the plant. This prevents colonization with harmful bacteria and speeds up the conversion of organic fertilizers. A special advantage that LABs offer is the conversion of sugars to hexanoates (salt of caproic acid = hexanoic acid shown in source 4), which are precursors and building blocks for terpenes. This means that the plant does not have to produce these substances itself, which is energy-intensive, but can use them directly. This saves energy, which can be used again for the production of sugars and other metabolic processes.
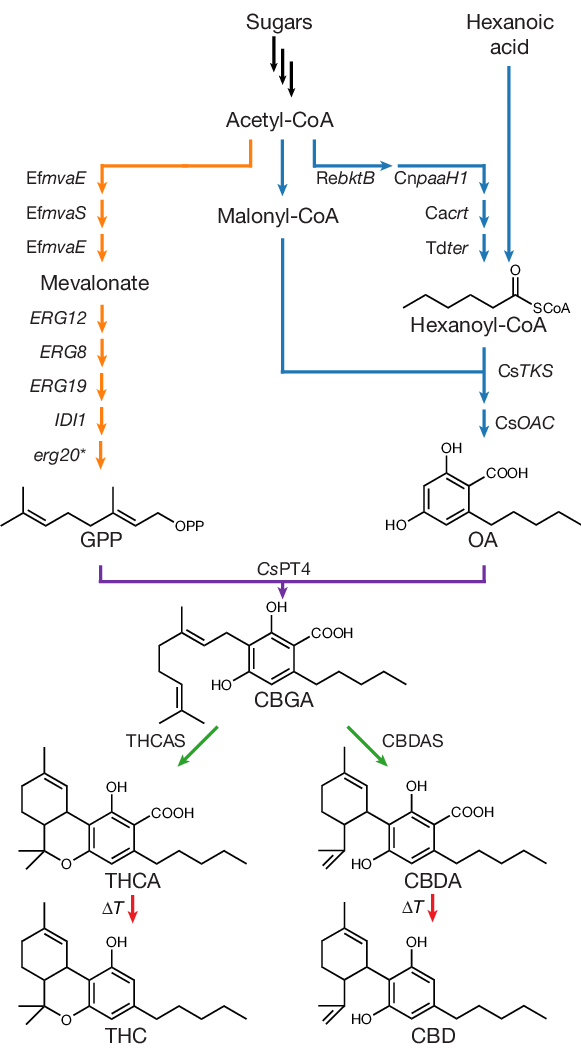
The next and important point is the use in the phytosanitary field (pest prevention). Again the LABs can shine by being extremely strong and competitive against other microorganisms. As already described in the production, after a certain incubation/propagation time they can take over almost the entire area by claiming the available food (sugar, especially lactose) for themselves.
Not only against other bacteria but also against fungi such as powdery mildew they can successfully assert themselves. This is how the oldschool trick of the milk/water mixture against the uninvited guest came about. However, this is now much more efficient and faster with the help of our LAB serum.
The concentration for this also remains at 1:1000, since the LABs can multiply very quickly, if enough food is available. The whole thing should be applied as a foliar spray by spraying the plant with an atomizer, but take care not to go below a particle size of 5µm, otherwise this will kill the bacteria.
The treatment is extremely effective in a protective (preventive) way, but its effectiveness decreases with the severity of the infestation, so it is only partially curative (plant is infested but does not show symptoms yet) and poorly eradicative (strong visible infestation). In case of eradicative planned action, a synthetic fungicide should be resorted to, if it is really absolutely necessary to save this cultivar as in the case of phenotypic selection.
Another point to consider with lactic acid bacteria is their ripening delaying effect, which can give undesirable results especially in the last 2-3 weeks of the flowering phase. From week 3 at the latest week 5 (depending on the duration of cultivar flowering of course) the addition should be stopped, otherwise neither the calyx hairs will turn brown nor the trichomes will mature properly.
Use as compost starter
Now we come to one of the most important point of LAB application: Its role in the initial processes of any compost.
First, we should look at the basic process of lactic acid fermentation, which is an anaerobic (without oxygen) catabolic (degrading) process. Here, glucose is converted to pyruvate and this is converted to lactate (salt of lactic acid) with the help of a coenzyme. This lactate is the starting material for the following decomposition processes, which finally leads to our beloved compost.

This process can be further divided into homo- and heterofermentative processes, after which the respective bacterial strains are also differentiated.
The first process is the "pure form", which means that this degradation is pure, i.e. apart from lactate no other by-products are formed (apart from the "used" coenzyme). Although lactate is also acidic, it is much gentler for microbes than acetic acid, thanks to its lower pKA value. Because with a low pKA value, the acid or salt of an acid can interact poorly with the microbial cells.
Heterofermentative fermentation, on the other hand, as you can probably imagine, produces other substances such as acetic acid and ethanol, which are highly toxic to our compost. This can lead to a lowering of the PH levels and the subsequent tilting of the population. You can recognize this at home by a strong smelling compost, which looks rather slimy and structureless than decomposed.

The starting point of these microbial conversion processes in compost is almost always started by two different strains of LAB.
The first is "Pediococcus acidilactici" (homofermentative), which accounts for more than half of the bacterial strains grown, this inhibits the synthesis of acetic acid, which in turn is extremely toxic to most auxiliary bacteria.
The second one is "Weissella paramesenteroides" (heterofermentative), which is the counterpart of the above mentioned specimen. This one produces exactly large amounts of acetic acid and slows down the composting process accordingly. This synthesis drastically decreases the PH in the entire colonized area and thus prevents the start of the decomposition process.
Accordingly, one should pay attention to the ratio in which it is applied. In a study of the "the science of total Environment" Magazines a ratio of 10^1,5 was found as optimum.
In this optimal ratio, the lactate blocks the PH reduction by the acetic acid and thus allows the colonization of the rotting raw material with fungi, which can degrade complex organic compounds such as lignin or chitin. The resulting degradation products again form reactants (starting materials) for terpene synthesis and activate systemic (plant internal) pest defense processes such as the formation of R-proteins.
If these fermentation processes are now finished after approx. two days, Paecilomyces species begin with the colonization of the material, the degradation of organic acids (PH increases -> aerobic bacteria settle from approx. Ph=6.5). Thus, the microbial aerobic degradation process can be started.
Combination with other KNF products
In the last part of the application in the horticultural field, the synergetic effect with the other KNF preparations must of course be mentioned. Mostly it is used right from the beginning as part of the Seed-Soak Solution (SSS), an organic solution that optimally prepares the seed for germination.
As mentioned before, LAB can repel competing bacteria or fungi (e.g. Pythium) lurking on the small, weak seedling. The exact mixture of the SSS will be discussed in a separate chapter, but roughly speaking, it is a combination of BRV (brown rice vinegar), FPJ (fermented plant juice), OHN (oriental herbal medicine) and our LAB.
But what are the other preparations for? Good question, because these also bring massive benefits to your sustainable garden. The rice vinegar buffers the PH range around your seeds into the right range so that the formation of anaerobic metabolites (alcohol etc) is suppressed.
The fermented plant sap gives a wide range of beneficial raw materials such as yeast species/hormones/enzymes and nutrients. This little lunch package helps the seed to establish itself quickly and strongly so that it quickly enters the "safe" phase where it can no longer be destroyed by just a small fungus.
Finally, the OHN is added, which together with the LABs plays the bouncer against fungi. The combination of alcohols (very very low concentration, which damages pathogens, but cannot harm our seed) and the active ingredients of ginger, angelica, licorice, garlic and cinnamon makes it almost impossible for pathogens to damage the seedling.
Another important application is the combination with IMO (indigenous microorganisms). Here LAB forms one of the starting materials in the collection of IMO also again with the protective aspect. Because we want strong, composting fungi and not fast-living bacteria.
But also in the preparation of a new soil mixture it can be added together with IMO or Liquid IMO to create a good base and stimulate the nutrient cycle.
| Inputs: | Tasks: | Mixing ratio: | Seedling: | Vegetative state | Flower I (Week 1-4) | Flower II (Week 5-8) | Flower III (Week 9-10) |
| OHN | Medicine | 1:1000 | 4ml | 4ml | 4ml | 4ml | 4ml |
| BRV | Catalysor | 1:500 | 8ml | 4ml | 8ml | 8ml | |
| FPJ | Food Veg | 1:500 | 8ml | 8ml | 8ml | ||
| FFJ | Food Flower | 1:500 | 8ml | 8ml | |||
| LAB | Starter | 1:1000 | 4ml | 4ml | |||
| FAA | Fuel | 1:1000 | 4ml | 4ml | |||
| SW | Minerals | 1:30 | 120ml | 130ml | 150ml | ||
| WCP | Bone builder | 1:800 | 5ml | 5ml |
How can I grow the LAB myself?
We have now heard about its many uses, but how do we put it into practice?
First, we need some materials:
- rice
- A lactose source (milk/milk powder/isolate)
- Clean water
- A large glass jar
- A breathable cover (paper (uncoated)/silk cloth/fabric)
- Rubber band/thread
- First, we soak the rice in water for 48std to filter out the starch. The rice should be completely covered and stirred 4-5 times.
- The water is filled into the previously washed jar to about 2/3, covered with the help of the cloth and tightened with the rubber band. This prevents pests from creeping in. It is especially important that the jar does not contain any residues of detergents/vinegar cleaners or similar agents.
- Now you can incubate the jar at room temperature out of reach of sunlight for 3-5days. The time varies depending on the temperature and richness of the environment in lactobacilli.
- After the waiting time, a semi-solid film should have formed on the surface. Do not worry it is not mold
 (Source 1)
(Source 1) - Now you can remove the lid and pour off the water, but be sure to remove the semi-solid part that floats on top.
- Then the lactose source, in our case milk, can be mixed with the opaque water in a 10:1 ratio. However, the final glass should still not be more than 2/3 full, otherwise it will overflow.
- The jar can now be sealed again as before in a breathable manner and stored. The mixture should now not be shaken during the fermentation phase.
- After another 4-6 days the mass should have separated as shown in the picture below. Again, the temperature is crucial, however, 25 ° C should not be exceeded, otherwise it can go bad.
 . (Source 1)
. (Source 1) - The yellow liquid is now our LAB serum, which we strain and take great care not to mix it with the solid again.
- The mixture should have a slightly sweet smell and if it starts to stink (sour milk), pour it away.
- If the serum is to last longer than 3-4 days it must be stored half-open (CO2 is formed) in the refrigerator. For very long storage (3 weeks +) it should be mixed with equal weight of brown sugar so that there is no unbound liquid left. This puts the LAB into a kind of cryo-sleep also called dormancy, where they become active again as soon as enough water is available again.
Can I still use the curd/lab?
The real rennet or curd, which has settled on top, can be removed and eaten. Especially for animals like dogs, pigs and chickens it is very nutritious and helps the digestion by its probiotic effect.
For human consumption, however, it should first be made into cheese, for this there is a super
tutorial video by Chris Trump
Lactobacilli and the human intestinal flora
As already mentioned in the introduction, lactic acid bacteria contribute significantly to intestinal health. Here they keep the balance between acidifying and alkaline acting bacteria. Because by fighting for nutrients, they can outcompete smaller, faster-digesting bacteria. This is good for us, because when bacteria convert nutrients, mostly H+ ions are released, which can lead to a rapid drop in Phs (PH = negative decadic logarithm of H+ concentration). This causes an unbalanced intestinal flora, resulting in digestive problems and even ineffective utilization/absorption of nutrients.
Unpasteurized dairy products contain a lot of lactobacilli, but can also contain pathogens so we would personally advise against it unless you have a microscope and can really differentiate the species.
A good option would be tiny doses of LAB serum (too much can easily lead to unpleasant side effects like diarrhea or abdominal pain). The best alternative in our opinion is to process the "curd" into cheese and eat it. But even here you should pay close attention to the quality, because the curd can sometimes tip over due to the non-sterile production, if it is too warm. This has a similar effect as fermented milk on one so it is not recommended.
But there are many good tutorials on cheese making and ripening with separate microbial strains, so you can easily make your own dream cheese.
Sources
- "Natural Farming: Lactic Acid Bacteria"; David M. Ikeda1 , Eric Weinert, Jr.1 , Kim C.S. Chang1 , Joseph M. McGinn1 , Sherri A. Miller1 Cheyanne Keliihoomalu2 , and Michael W. DuPonte2 1 Cho Global Natural Farming Hawai‘i, Hilo, HI
- "Lactic acid bacteria modulate organic acid production during early stages of food waste composting";Quyen Ngoc Minh Tran 1, Hiroshi Mimoto 1, Mitsuhiko Koyama 1, Kiyohiko Nakasaki 2Q
- "Lactic Acid Bacteria and Bifidobacteria with Potential to Design Natural Biofunctional Health-Promoting Dairy Foods"; Daniel M. Linares1,2, Carolina Gómez1, Erica Renes3, José M. Fresno3, María E. Tornadijo3, R. P. Ross2 and Catherine Stanton1,2*
- "Complete biosynthesis of cannabinoids and their unnatural analogues in yeast"; Veronica Benites;
- "The value of Lactic Acid Bacteria in Bees’s stomachs and honey for human medicine"; Mark;
- "Biotechnological valorization of agro industrial and household wastes for lactic acid production";Juliana Romo-Buchelly, María Rodríguez-Torres, Fernando Orozco-Sánchez;
Korean Natural Farming I The future of sustainable agriculture
Why should I use KNF?
Our environment is not doing well, whether it is due to climate change, overuse of pesticides or over-exploitation of fertilizers.
One major influence is the large monocultures that make up a big part of the crops grown annually around the world. In the graph below, we see that corn accounts for almost a third, the majority of which is used for animal feed.
This intensive land use has resulted in an enormous removal of phosphorus from the soil. This, coupled with a steadily increasing world population, poses a challenge to conventional agriculture which will not have enough mineral phosphate fertilizer to meet global demand by about 2050. Below we have included a small comparison between phosphorus supplies and its consumption.
The occurring shortage of phosphorus also has the effect that there is less yield per plant. This again increases the demand for agricultural land, which is decimated by the increasing sealing of areas. Now the question is: How do we combat this trend?
One promising option is to combine conventional methods in a mitigated form with a sustainable, regenerative farming style such as Korean Natural Farming. Especially by using indigenous adapted microorganisms and the very effective composting/recycling of organic waste, we can counteract this trend.
It should be clear, however, that on a large, world-supplying scale, not only natural farming methods can be used, since one must ensure a basic security or basic yield with fertilizer salts.

However, we as cannabis growers and "small scale producers" can fully work with these methods and thus reduce our Co2 footprint. This is because our beloved indoor growing method in particular is unfortunately very resource intensive.
While this is usually essential for legality reasons, KNF and Living Soil methods can save quite a bit.
How to do this and still keep your yield or even increase it, we will take a closer look at in the next articles.
What exactly is Korean natural farming?
The origin of this concept can be found in, as the name suggests, Korea. South Korea to be exact. Here the inventor Cho Han-Kyu, also called Master Cho, thought about how to achieve good results in horticulture/agriculture with old techniques, implemented in a modern way. Through this, a holistic concept was created, which owes its extraordinary results to the interaction between indigenous microorganisms and fermentation processes.

Special attention was also paid to cost minimization. By using locally available inputs and waste recycling, KNF is one of the cheapest methods to achieve good results.
Master Cho got the basic ideas behind local inputs from his studies in Japan, where he spent several years before that with highly respected horticulturists such as Yasushi Oinoue.
Back in South Korea, he combined this with the "old" techniques of the Koreans, who had already done some research in this area through kimchi and other fermentation products.
This resulted in the concept that is now trending among sustainable farmers worldwide.
It works so well that Master Cho has already been arrested due to pressure from agricultural companies in South Korea and has been imprisoned for a short time. But his teachings were still spread around the world and eventually he was released again.
How do we make use of the full potential?
As described in the previous chapter, KNF works by combining different preparations that perform different tasks.
This division makes it possible to create the perfect mixture at each stage of growth. But what is absolutely necessary, what is optional and how do you obtain your preparations?
This and much more will be described in more detail in the coming articles. This article will give you the basics of Korean Natural Farming so that you can choose which mixtures to apply at which time.
The Basics
We have already said several times that the interaction between the living organisms and parts of the soil plays the most important role in KNF, but why not just take bottles from fertilizer manufacturers, which promise the highest possible yield with their mixture?
Although these can also give good results, the labor and environmental aspect is crucial here.
To get the same yield and quality standards on a hydroponic system as on a Living Soil, you have to grow the same clone more often to meet and not exceed the respective nutrient requirements.
This is because mineral nutrients actually mean nothing other than that they are already present in their charged (ionized) form. Nitrogen, for example, is present in fertilizers as nitrate (NO3- for annual plants) or ammonium (NH4+ for perennial plants). Through the charge, the nutrients are more or less "pressed" into the roots, as substances are absorbed here via charge gradients.
Many compare this procedure with force feeding, because the plant has no chance to reject excess nutrients and thus the famous "fertilizer burns" occur.
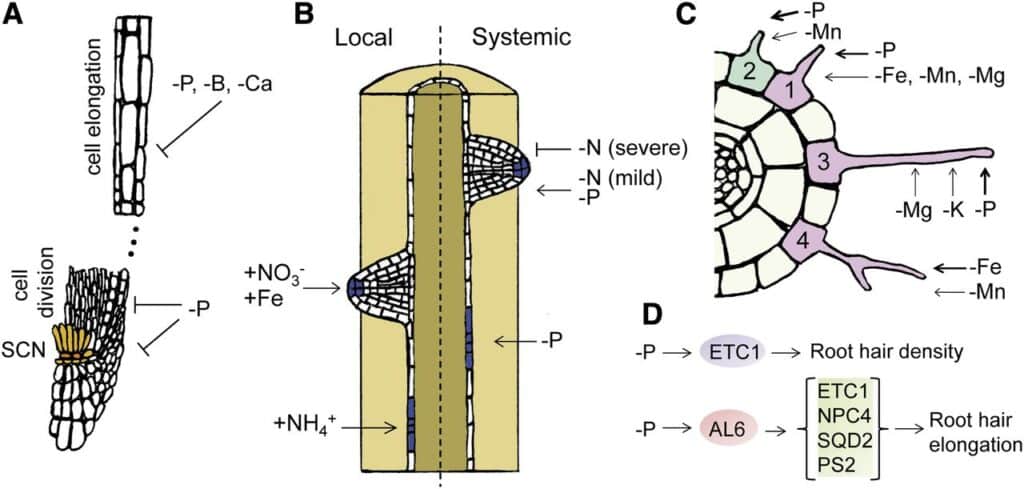
In so-called organic cultivation, on the other hand, one uses the symbiosis between plant and microorganisms/fungi. It is true that the term "organic" is difficult to define, since rock flour is also used here to supply minerals. These are partially ionized and therefore immediately available.
However, the majority, just like other inputs, is mineralized only gradually. How fast this works depends on the mineralization rate, which in turn depends on many factors.
I will break this down in detail in the Living Soil article, but here I have written down a small list of influencing factors.
- PH-value of the soil (6-7 is best)
- Temperature (20-25°C is optimal )
- Microorganism composition
- Microorganism quantity
- Soil moisture
- Soil structure
All these factors can be positively influenced by adding KNF product. These either directly promote the microbial population, provide nourishment or displace pathogens (harmful organisms). The soil structure is also positively influenced, as the bacteria release a kind of slime that cements the soil particles together, thus creating better water-holding properties and nutrient storage.
These microorganisms can be added exogenously (from outside) to push mineralization in a certain direction. However, this is not an instant solution; the goal should always be a balanced population of different species.
In fact, if you have a complete biotope, the plant controls the amount of certain bacteria through its root exudates. Root exudates are sugar compounds on which microbes can feed excellently. The plant exchanges them for nutrients, which in turn promotes the particular microbial species that provides the desired nutrient.
A small example: The plant wants more potassium, then it releases a specific exudate matrix, which is detected by the potassium-releasing bacteria and these are then stimulated to exchange as shown by the illustration.
Furthermore, VOCs (volatile organic compounds) and organic acids are released for pathogen defense and release of nutrients.
The importance of local Inputs
Why do I talk about local inputs all the time? I know I sound like a broken record, but this factor has a serious impact on the success of KNF products.
We are taking advantage of the full range of organisms that are perfectly adapted to our location. In other words we use natural selection to find out which microbes are best suited for our spot.
We also protect our native biotope, which would otherwise be displaced by sometimes invasive species. While this may be partially intentional it breaks down the natural homeostasis (balance) and can lead to undesirable side effects.
Another point is that the plant can develop to its maximum potential by controlling its own nutrient supply. This refers not only to yield but also to the quality of the material such as terpene content and trichome number or density.
This does not mean that locally there are only good varieties. You should follow the recipes exactly and pay attention to identifying characteristics like color and odor. How to determine these exactly I will show you at the respective products.

If you are interested in the philosophy and further insights into the development and application of KNF, you should read the books of Master Cho himself. Some very good Youtube videos are also available from KNF greats such as Chris Trump or PureKNFDrake.
The Life Cycle of Plants
Now we have learned that with different inputs you promote different populations which again have an impact on the control of the plant. Now let's take a look at the life cycle of a cannabis plant and what is needed in which phases to be able to compose the different recipes.
Seedling/Clone
In this phase we don't need strong nutrient products yet, but we just need to prepare the soil and the seedling for the coming growth phase. For this we provide good microbes and prevent the expansion of the population of pathogens.
Vegetative Phase
In this phase the foundation for a successful harvest is made and we have to feed the plant accordingly. Products with very high nitrogen or amino acid content should be given together with the basic supply. This is where the root system and a stable branch/leaf system establishes itself. That is why we should apply both soil and foliar fertilizers.
Flower initiation
We now have a strong and thick growing plant and want to move into the flowering phase as quickly as possible. This should not only be fast but also corresponding with the yield potential.
To create the right basis for a high yield, the plant needs the basic supply and especially a boost of calcium and phosphorus in this phase.
Main Flower Stage/generative Stage
As the name suggests, this is the phase in which most weight is gained and quality can be significantly influenced.
Special attention should be paid to the supply of minerals and potassium/phosphorus.
It is also important to mention that we work here only with soil application, because we do not want to have residues of the agents or a risk of mold (Botrytis cinerea).
Ripening
We enter this phase in the last 1-2 weeks before harvest. Now we want to induce maximum terpene formation and maturation of the trichome heads.
For this we use similar inputs as in the main phase
A brief overview of KNF products
Now you are probably wondering, what should I use in each phase? The answer will probably be superfluous after this chapter, because I will now explain to you the main components of the KNF regime.
Here, however, it is not possible to distinguish by a strict NPK specification as with conventional preparations, because the products are so much more than pure fertilizer salts. In order not to go beyond the scope of this short overview, I will deal with production and exact modes of operation in the respective articles on the specific products.
For this reason, KNF tends to speak of tasks that the product performs. For OHN, for example, the term medicinal component is used because this strengthens general plant health.
Structure: Abbreviation = original name written out = translation = effect.
OHN = Oriental Herbal Medicine = orient. Herbal Medicine = medicine -> general plant health is strengthened by stimulating the immune systems
BRV = Brown Rice Vinegar = catalyst -> without this the PH partly fluctuates and some other inputs can not be implemented
FPJ = Fermented Plant Juice = food -> nutrients broken down/available from green plant material through fermentation + carbohydrates for
microorganisms
FFJ = Fermented Fruit Juice = food -> same principle as FPJ except that here fruiting parts are taken and therefore in the flowering phase
Application
LAB = Lactic-acid Bacteria = Lactobacteria = support -> these very strong microorganisms displace pathogens, fight Botrytis and accelerate
composting
FAA = Fish Amino-acids = fish amino acids = fuel -> this preparation gives especially in Veg really gas by the immense N-content and the completely preserved
amino acids the soil life is stimulated so strongly that the soil temperature rises and the plant makes large
growth leaps in a short time
IMO = Indigenious Microorganisms = backbone -> IMO makes up to 80% of the KNF performance and is therefore the most important ingredient, since it
brings the basic stock of soil life without which nothing works. Here there are some
gradations, but we will only go into this in the designated chapter
WSCP = Water-soluble Calciumphosphat = Bone-soluble Calciumphosphate -> Hereby we support the plant in the build-up of flower buds through
additional calcium and phosphate. This allows more nutrients to pass through the
ER (endoplasmic reticulum) and thus a better supply can be guaranteed.
supply can be guaranteed
SW = Sea Water (water + 5% salt) = Mineral Complex -> This rather simple preparation consists of only two inputs, but has a strong influence. Salt is extremely
full of nutrients and should therefore be used sparingly.
How do I combine these Inputs?
Now the question is what fits best in which stage. We have already outlined it in the description of the stages, but we still have an exact list for you here. This is the compilation as master Cho personally created it for 4L of water
| Inputs | Aufgabe | Mixtureratio | Seedling | Vegetative | Flower- Initiation | Main Phase Flower | Ripening |
| OHN | Medicine | 1:1000 | 4ml | 4ml | 4ml | 4ml | 4ml |
| BRV | Catalyst | 1:500 | 8ml | 4ml | 8ml | 8ml | |
| FPJ | Food | 1:500 | 8ml | 8ml | 8ml | ||
| FFJ | Food | 1:500 | 8ml | 8ml | |||
| LAB | Supporter | 1:1000 | 4ml | 4ml | |||
| FAA | Fuel | 1:1000 | 4ml | 4ml | |||
| SW | Minerals | 1:30 | 120ml | 130ml | 150ml | ||
| WCP | Bonebuilder | 1:800 | 5ml | 5ml | |||
From this you can already deduce it: The standard administration in each phase consists of the so-called "Maintenance Spray", which includes OHN, BRV and FPJ except in the ripening phase, here you replace the FPJ with FFJ from ripe fruit. From there you can see what the plant needs or what would still be beneficial and can then add it on top of the Maintenance Spray (MS).
For example, if there is a calcium deficiency at the beginning of flowering, you add WCP to the MS and use it as a spray.
Now we only need the recipes and seasonal tips, then we have also worked through the chapter KNF.
One tip in advance, store a lot of brown sugar and by that I really mean a lot. You will need it for the next recipes.
If you like our articles and want to stay informed about competitions, events and our passion for writing: we are also available on Facebook, Linkedin and Instagram.
Sources
- „Korean Natural Farming: Master Cho Biography“; Nico Hill for Gardenculturemagazin; 06.05.2019; (https://gardenculturemagazine.com/korean-natural-farming-master-cho-biography/)
- „Cho’s Natural Farming: Recipes and Instructions for Use“; Cho Han-Kyu
Illustrations
- Pie Chart; VOX; (https://www.vox.com/a/explain-food-america)
- Maize P Mangel; Mary; (https://www.pinterest.nz/pin/38632509278445644/?autologin=true)
- Improving Plant Phosphorus (P) Acquisition by Phosphate-Solubilizing Bacteria; (https://www.researchgate.net/publication/318963768_Improving_Plant_Phosphorus_P_Acquisition_by_Phosphate-Solubilizing_Bacteria)
- Root Nutrient Foraging; R. Giehl, N. v. Wiren; (http://www.plantphysiol.org/content/166/2/509)
- „A Return to the Wild: Root Exudates and Food Security“; C.Preece, J.Penuelas; (https://www.cell.com/trends/plant-science/fulltext/S1360-1385(19)30247-X)
- „KNF and IMO“; Nico Hill; ( https://gardenculturemagazine.com/korean-natural-farming-and-indigenous-microorganisms/ )
Ice water hash - a complete guide from harvest to dab
The amazing title picture was taken by the one and only Even Stone. Follow him on Instagram!
What is water-hash?
As you probably already expected from the heading for this method of separating trichomes from cannabis flower, we use a combination of ice and water. This mechanical procedure uses the force of the water vortex to brush of the oleoresin containing trichomeheads from yours buds. The ice in ice-water-hash making is only used to cool the whole mix down in order to make the stalks of the trichomes brittle so that they break of more easily. But the exact steps you need to take and much more will be discussed in the following article.
Why choose water-hash?
When you hear about ice-water-hash most times it sounds pretty labourintensive and it is, but the ratio of quality per worktime is the highest of the different solventless methods. Dry-ice-sifting for example oxidises the trichomes so fast that they burst and leaks out the desired compounds like the really volatile monoterpenes (limonene). And not only the trichomes burst, the plant material gets also really brittle, really fast so the contamination rate also increases rapidly. This results in a drastic loss of quality and worth, because even when pressed this can only achieve low prices on the market.

Then why not use drysift, there's no dry ice used and no enhanced oxidation. Thats completely true, but as we're going for most quality per time drysifting also loses in that category for it's time intensive cleaning process of the material to get to 90% purity. Don't get me wrong I love drysifting and such fire as this fullmelt drysift from CubanGrower x The Cuban Hash Queen (Pheno by TheVillage/Photographed by Erik Nugshots/Bred by Symbiotic-genetics)makes it worth it.

But to be profitable with that kind of premium product is really difficult and the market is not that big. So in order to sell at a lower price, but still nearly the same high quality as with drysift only ice-water-hash remains an option. "Why not just blast it all"?
Good question, and yes you could do that, but the price for bho currently is not as high as decent hash rosin. Additionally the startingcost is really high if you want to do it right. If you need deeper info on hydrocarbonextraction and it's specifics, visit Murphy Murris Instagram account
And for home users I wouldn't recommend it either if you don't have a proper, safe setup and are quite experienced with hydrocarbon handling. Hash on the other hand doesn't need a C1D1 room or other security measurements than a rubber mat on the slippery floor.
So if you want to make fullmelt the fastest way possible use ice-water-hash to separate your resin like the italien hashmaker slite23 from Barcelona with this beautiful Sundae Driver 149/90u Fullmelt (Pheno by Alorganics). Now we will look a bit further into the mechanics behind hash-making in order to build a good knowledge base, that will let you achieve resin like this with a bit of practice.

The principles of Hash-making
Terminology
In the first part of this chapter we will look at some basic terminology that we need to describe the process precisely
- Micron: This term refers to the opening space in the bubble bags. It tells you how many micrometers the wholes in the screen are wide. These mostly get abbreviated as "u" or "μ". To put that into perspective, a human hair is 75 micrometers thick and our eyes can only see down to 40 microns. Thats why it's important to buy a scope and look at the holes in the screen to see if they are all open and not clogged with nylon or bad stitching. Always wash your bag properly before using them. For washing the first time after buying new bags, I would recommend to make a 10% alcohol/water solution and rinse the heavily. But be careful not to rip the seams.
- Terpenes: These pure hydrocarbons are a part of the famous entourage-effect firstly mentioned by Dr. Ethan Russo in his paper "Taming THC: potential cannabis synergy and phytocannabinoid-terpenoid entourage effects" (see source 1) They induce a synergistic effect in combination with Cannabinoids in your brain, basically pushing the high in a certain direction. Like Limonene for example, that on its own already causes a sort of high if you breath it in at a certain concentration. Similar to that strains with high linalool make you relaxed and the same for other strains with a lot of terpenes. There are also other components like thiols and alcohol esters in Skunk e.g but they aren't as well researched as terpenes At last there are also terpinoids, which are similar to terpenes in effect and structure, but the have additional chemical groups. But because of their similarities we mostly count them to the terpene content.

- Fullmelt: We already used this term in this article and some of you probably wondered what that means. It stands for the quality of the hash in regards of melt % tage. Hash that melts like water in your banger and nearly leaves no residue is the best of the best in hash. At room temperature it sometimes already melts into a oil like mass of trichomes. More info on what differentiates the melt further and how you classify it will be discussed in the chapter Rating-Systems
- Contaminants: All particles that aren't trichome heads be it hairs, skin, dust or plant particles are considered contaminants. More on how to prevent them and proper hygiene in the coldroom in the designated chapter
- Cultivars: Every plant that is derived from sexual reproduction is, like humans, different even from his siblings. This term refers to one unique genotype with a specific phenotype that gets expressed. So for example just because some Cake clones yield not all of them will and even if you have a selected cut it might not dump a lot of weight if grown under completely different conditions like those that it was selected in. That's why it's good to ask the clone seller how it was selected or select your own cultivars
- Flowrate: Like in coffee brewing you can control some parameters like speed of water drainage and with that force that pushes through the screens. Using that you can safe time by not having to spray and rinse that much. But that needs the perfect speed and that takes practice as you have to be careful not to blast the water through as that could drag contaminants through the higher micron screen and also degrade your trichomes by removing more terpenes than necessary. But not enough pressure prevents the contaminants from getting flushed through
- RO-Water: Growers already know the term, but for everyone else: it refers to water filtered through a reverse osmosis filter. This device pulls out all the minerals and other compounds that could affect the taste. For optimal results you should make your own RO-Ice cubes (Protip: Insulate your washing vessel to save ice and prep time)
The Separation Process
How exactly can you isolate the trichomes without plucking them off one by one? For that to work we need Physics. Trichomes are secretion-organs with bulbous, oleo-resin filled heads on them. The oleo-resin is trapped by a layer of lipids that releases terpenes and other compounds slowly over the day on the plant to fight of pest. It also shields the flower from UV-Damage so to increase your trichome-coverage you should incorporate UV in your lighting mixture.
As we know from chemistry class, fatty substances don't mix well with water as they're non polar. This phenomenon is the reason why trichomes stay intact while beeing washed most of the time. If your strain as a to thin lipidlayer the trichomes smear in the bag and you won't catch most of them

That's why some cultivars won't produce hash yield numbers that justify washing the material. Even if it has a lot of good trichomes you have to separate them completely in order to catch all. Therefore they have to be evenly brittle, but not brittle.
Thats where water comes into play. This natural "solvent" stays, even with ice, at 0-5°C for the whole washing-process to insure maximum Quality and efficiency. Now that we have the Trichomes ready for action we will start the stirring process. If you're hand-washing try to get a small water vortex as the current of the water strips the flower from the Trichome heads.
If you picked a good cultivar the water should turn slightly golden/purple (depends on the strain) and you should start to see the heads drop to the bottom of the wash container. If you think you got most of them, you can stop agitating and let the trichomes settle down. After that you skim the water on top that doesn't contain resin and either filter it or throw it away. Let about 1-2/3s in the wash vessel and strain them through the Bubblebags.
How to collect the Resin from Water
But in which order do I have to put the bags in? We go from big to small openings but how many bags you have depends on your bag-set. Different companies also use different mesh sizes but most of them are in the same region so we want to give you a small List of bags that there are.
- 220μ = Filter-/Workbag: most of what's catched here is dust and plant material. This is to clean up most of the contaminants before catching anything. If you buy one that is big enough to fit into your washing vessel, you can wash in it and pull out most of the plant material even before emptying into the filter vessel.
- 180μ = Second Filter: Most bag-sets don't have them but they're a good option if you want to add another filtration process to your regimen. Most of the heads catched here also don't have a lot of value
- 160μ = Rosin-/Foodgrade: This bag collection is worth it, especially when you grow strains with really large trichomes like a GMO. You can even find some partial melt in here, but I would recommend to turn this into rosin or edibles/RSO as most of the time it is still a bit dirty.
- 120μ = great Quality Resin: This is where the real fun begins. In this range you start to see real fullmelt or at least 4 Star quality, that is either worth keeping as hash or if it's doesn't melt fully to press it and sell it for premium prices
- 90μ = exceptional Melt: The melt coming out of this bag is the true Creme de la Creme and goes over the counter for astronomical prices up to 130$/g. The fullmelt nearly leaves no residue on the banger and the taste is amazing. As said before, all non melt result should be processed to rosin or oldschool hash (Frenchy Cannoli-style)
- 70μ = is used more like a substitute for the 90μ bag. To test which one you like better, you should test wash and compare how the cultivar performs with the different bags. Most intact, ripe Trichomes are not smaller than 70μ so this will be the last bag with exceptional results.
- 45μ- 25μ = Foodgrade: This bag catches some debris, but mostly unripe or already dried up trichomes so it can be used similar to the 160μ
- "Full-Spectrum": This term is a tricky one, because a real full-spectrum Oil refers to it containing the whole range of cannabinoids and terpenes. But most Water-Hash fullspec is a 45μ through a 160μ or similar micron sizes. This leaves out a lot of other compounds so technically it's not a fullspectrum oil in my opinion. But as with everything I teach, I would be really happy to discuss these topics in the comments. Real fullspec is only achievable with solvent-extraction as it can extract everything contained in the flowers.


What do I do with the wet Hash now?
Now that you catched all your trichomes, you want to make them smokable by drying them really thoroughly to prevent it from molding. This can happen in the dry stage aswell so be fast with the process. There are two main options for drying Ice-water-hash. The first and cheapest option is air drying it by freezing it immediately after collection and spreading it out really really fine after that. How to do this and the exact steps will be mentioned in the drying chapter.
The most used method today is the use of a lyophilisation ofen also called freeze dryer. This neat piece of equipment uses certain physical phenomenons to quickly remove the moisture from the trichomemass. It also is the most terpene and colour preserving technic for drying. For commercial ops this device is essential for production speed and success as a failed air drying attempt could cost you a lot of money.
How to prepare yourself before setting up a hash lab
Now most of you probably would want to immediately wash their whole crop, but before doing that, we need to set up a proper lab and everything that goes with it. For starter we need the most essential item to be of the highest quality possible. Of course I'm talking about the starting-material as it is our base for good hash. That's why the mantra for most hash makers is: "Fire in, Fire out" To give you an idea of what I mean with that, we will look at our material in depth and disect what makes good Hash starting-material
The different aspects of good startingmaterial
Live vs cured Flower
The first choice with have to make after harvest is whether we want to make "live" or "cured" hash . These terms refer to the drying process of our material. Live Hash is taking the fresh flowers and directly freeze them for washing. That way, you can conserve the most terpenes especially those that flair of easily like monoterpenes. This brings more sharp, intense flavor that many people nowadays really enjoy. also the colour gets "better" more easily with this method as nearly no degradation happened. What we mean with better and which role colour plays in hash, can be looked up in the designated chapter With this method it's vital to have an extremely clean freeze, if possible you should have a separate one only for fresh frozen flower. But if you only wash once every 2-3 Months this isn't really worth it, so you can use tupperware to protect your buds from the other freezer content.

Often the whole plant gets frozen for this purpose so you will sometimes see the term WPFF on your bought hash. This stands for "Whole plant, fresh frozen"

Cured Resin on the other hand has a more "rounded" flavor as the highly volatile Monoterpenes have flaired of or turned into another terpene profile. Most of this is still in research, but you definitely taste a difference. This rosin is bit harder to get as light in colour as the live version, because the dry time has to be perfect. But as we will see in the further chapters, colour doesn't always say much about quality.
How to visually rate Material
Now that you picked your methode of choice, we can start to analyse if our product will be cut out for making hash. As mentioned before the trichomeheads and their size play a mayor role in it.

First of all lets look at this depiction of a trichome. This shape is what you're looking for in the perfect trichome category. It has a large bulbous head with a really thin "neck". This is what we want as it will make it easier for us to break them off and separate from the plant material. What we don't want are secretory glands without heads, their "hairs" may look frosty af on a plant, but if you wash them, you will be really disappointed. A typical strain with these attribute is purple punch. It looks fantastic but washes really bad most of the time. Another unfavourable trait for hash making are varieties with really long trichome stalks, small heads and a thick waxy cuticle as they won't contain a lot of active compounds. These strains are also valuable, but just not for water hash.
Don't get me wrong, if you select a fire cut, it can dump, but you would have to go through a lot of cultivars to find one. I recommend 20-50 regular seeds depending on the breeder. You can do smaller hunts, but the probability of finding a keeper cultivar that satisfies both quality and quantity aspects will be quite low.
The importance of hash specific breeders
This is where the breeders come in. If you want to select a hash-cut but don't have the time to sift through hundrets of plants, you should pick a breeder that selects for hash. That will give you a lot higher starting chance of finding a good specimen as the breeder has already pre selected the genetic material. There are lot of different great breeders and most probably I will forget to name some and I'm sorry if I left someone out. But to give you a direction, here are some breeder recommendation from my side: Karma genetics, Oni seedco, Bloomseed co, Dying breed seeds, Archive Genetics, Truecannabliss and Cannarado to name just a few.
Some strains especially are good for hash-making as they have a high rate of successful cultivars. Names like GMO, Sour diesel crosses or the famous Gorilla glue are well known for their ability to dump wpff yields upwards of 5%. Even most crosses with there lineages will give you your desired results so they're a good starting point for your hash-journey. For companies I would also recommend to implement a really heavy yielding strain at all times in their rotation as a safety net, if some new selections perform worse than expected.
Now that we have picked a good strain, we can start growing and watching it. When the heads are nearly finished you can scope it with a jewelers lupe or a electronic microscope. This should be done extremely thoroughly because if you don't do it and do a test wash, you potentially waste money and thats not good when you're starting a company. To give you a comparison chart we depicted one picture from a study by Dr. Potter
Figure D depicts the best usable trichomes as we don't need excessive force for separating these from the stalk. Trichomes like in figure C aren't the optimal, sure they have a large head and contain more oleo-resin than the ones in figure B, but they require so much force to get separated that there also is a lot of potential for contaminants to get into the product. All these criteria are only the beginning of a long r&d (research and development) journey that should look at terpene-make-up, terpene Content, rosin yield, stability, colour etc
The distribution of Trichome size
Another important performance indicator is how much a strain dumps in each quality area. A cultivar that puts nearly all its resin out as fullmelt is much more valuable than one that produces 70% edible grade hash. As with every property of hash that we look at, we need to do our tests here as well.
For this purpose you should use a register/excel sheet to get an overview of your data. This data is a treasure chest of usefull information about your process, that can give you the opportunity to optimize every little detail. This would be a example of how to analyse a strain:
- Terpene make up
- Terpene content
- Yield WPFF vs Cured
- Market value
- How long is the overturn time (harvest-harvest cycle)
- Trichome Size

How to test your cultivar for yield beforehand
To test and find the perfect cultivar is quite a challenge so I highly recommend keeping a cut from said plant. Not only for hash making, but for sharing /selling the clone. The high end concentrate marktet is really hot right now and new, good clones go for a lot of money. So this would be the perfect example for horizontal expansion of your production line.
But to get such a cut, we have to select and before selecting we should do some tests. The first is a cannabinoid and terpene test which you need regardless as a commercial cultivator. If you buy flowers you should ask for their Coa so that you can compare your yield to the given data and check if you catched all the trichomes. This is also a great opportunity to fine tune your normal washing processes. So if you don't get the same % in yield that is given in the testresults you should go through your process step-by-step in order to find the mistake. If you don't find anything, it could be the strain. Look at how the resin handles the water and if it smears the bags. Most times this is a large opportunity for product loss.
If thats not the problem, try using the leftover material for edibles/rso, that way you can use every little bit of the material and minimize loses.
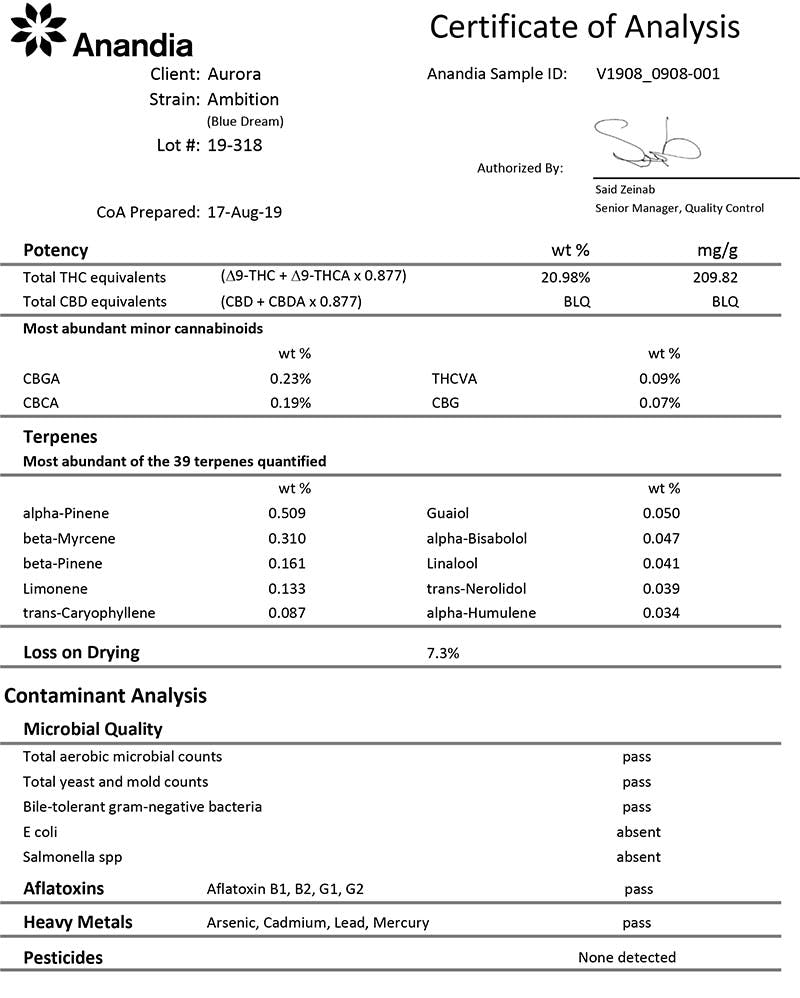
Another test is the mason jar shake. This simple method has a bit more to do with experience, but is also usable for beginners. You simply take 10-15 grams of your desired flower, put it in a mason jar together with Ice and water. After letting it sit for 2-3minutes you can swirl it around a bit and then you should see golden trichomes dyeing the water golden. Let it sit for another 2 minutes without stirring and look at the bottom of the jar. There should be a small layer of trichomes if you have a fitting cultivar.
Cultivation Techniques for better Hash

The best way to get great hash yielding flowers is to grow them yourself, so that you can learn the subtleties that go into growing the best hash flowers. One sure way to increase resin content is adding UV-lighting to your grow as we mentioned before. This lets the flower build up its defences against this high energy radiation. Not directly for resin content but for terps, most hash cultivators use organic living soil system to grow as this lets the plant fully express its genetic potential. This debate is highly discussed in the hash community so I don't want to get too deep into it. But it has to be said, that salt based fertilizer can also achieve great hash if they're perfectly tuned in for the cultivar. A general booster for terps and secondary metabolites is a healthy amount of sulfur in our regimen. Organic growers can use gypsum or epsom salt for this purpose
The necessary Equipment for Washing
Now that we know our basics and how to choose material, we need to look at our setup. To configurate it the best way possible we need to choose between the two kinds of washing. A pros and cons list should be sufficient to give you an idea of what you want to choose
Hand vs Machine Washing
Pros washingmaschine
- Multiple maschines can be operated by one employe
- Less manual labour
- simple SOPs can be made for unexperienced operators
- "easy" for beginners
Cons washingmaschine
- Lots of work to keep perfectly clean
- High initial investment cost
- cheap maschines need a draintube change in the beginning
- often not very customizable
Pros Handwash
- Easy to clean vessels
- Fully customizable washing program
- Really gentle on the material
- Smaller initial investment
- Large batches of 10-15k grams possible
Cons Handwash
- hard physical work
- Not automated
- Experience is needed to get perfect results
The next big decision is whether to air dry or freeze dry. But it would blow up this chapter so we will look at this in the specific chapter.
A short Equipmentlist
Now for a better overview we collected a list of small and big items every wash room needs in order to function properly.
- Bubblebag-Set (+ Workbag)
- RO-Water
- Ice-cubes (Ro-cubes if possible)
- Washing Device (Maschine vs Vessel)
- Stiring device for handwash (stainless steel paddle)
- Drainvessel for filtering the hashwater (should be big enough to mount the bags in it)
- Thermometer for water temps
- Extremly clean work enviroment
- AC for cooling the room
- Cleaningagent (isopropanol 70% works best)
- Waterhose with RO-water to spray down the bags
- Cold Spoon for collection
- Dryingdevice (Freeze Dryer or Microplane/Sifter)
How do I prepare my tools?
The most important factor is cleanliness, I know I sound like a broken record, but as everything gets concentrated with hash making, the dirt also gets concentrated. Thats why the phrase: "cleanliness is next to godliness" fits perfectly for hash making. You should start with your wash room and wipe everything down with a 50/50 alcohol/water mix. Because if you only clean your tools and put them back into the room, they get contaminated aswell. Even the ceiling and the walls should be cleaned as dust can settle there.
Next we need to take a look at our tools. They also should get wiped down and put in a cold place as the resin won't stick as much then. Your Bags should also get washed with a very mild alcohol/water mix and then rinsed with RO-water. After that you can put them in the drainvessel over night in order to dry them.

If you make your own ice you can make it the night before with RO. The best shape in my opinion is half round about the size of your thumb. This minimizes the surface and thus lets your ice stay longer like this.
How to keep proper hygiene in the coldroom
We explained how to clean your room + tools, now we go to the steps you can take yourself. The biggest vector of contamination is the human, so in order to mitigate this risk we wear the corresponding PPE (personal protection equipment) Lab coats are a standard in most hashlabs but a full-body tyvec-suit would be more appropriate for this usage as it contains the dirt on your trousers as well.

As you can see in the picture a mask and glasses are also a good thing to contain bacteria in your breath and skin pieces. But thats more important if you're in the medical sector. For home use you can get away with a suit, a hairnet and proper nitril gloves. These are really practical as your hands would get sticky really really quick without them

A coldroom buildout
Now that we protected ourself and the hash from contaminants we need to build a coldroom that makes our job as glorified janitors easier.
The best working surfaces
The first thing we should direct our attention to are the surfaces. Every desk, floor and wall should be wipeable as they collect dust. The best option would be to use stainless steel everywhere because it can be safely sterilized heavily without showing any degradation. Another reason is the faster cooling off the room because of it being metal. But such coldrooms are really expensive and only for professional operators For the homeuser, I would advice to lay out paintersfoil on the floor and walls. Looks like out of the movie american psycho, but trust me it is worth it. Also it is quickly removed if some uninvited guests want to look into your flat

I would also clean the ceiling with a broom or a mob and some water if possible before washing. If you want to be really thorough, you could put in an air filter. You can use a normal carbonfilter similar to the ones in growtents.
How a professionally built shell for your coldroom
Optimal climate control
For washing and separating we need constant temperatures around 0-10°C to get the best results. Otherwise the hash will get greasy and really bad to collect. This is quite difficult if you don't have well insulated place to wash. That's why we recommend using cellars or insulated sheds for home users. If your growroom is clean and sufficient you can use it as well, but every room needs a strong AC to wash in the summer months. But how do we know which one to buy? This depends on your budget and coldroom-size. Most Acs are measured in BTU/H or Watt/H which can bei converted into each other. 1000BTU/H are equivalent to about 300 watts/h and how many of those you need can be looked up in the chart we made
| Space | Watts |
| Up to 30m2 | 2350 |
| 30-37,5m2 | 2500 |
| 37,5-42,5m2 | 3000 |
| 42,5-50m2 | 3500 |
| 50-60m2 | 4000 |
But not everyone can afford the best Ac so we're going to look at some models for each price class
Small to medium budget
In this category we have the well known mini splits, which most of you will have in their grow room. This modell is really efficient and in my opinion the best choice for small to mid sized ops as you can get down to freezing temperatures in a medium insulated place because it doesn't use air, but a refrigerant. They're powerefficient and don't take up to much space, but it needs a second device on the outside of your building which has to be connected via copper wiring. That and the cleaningafford are the only negativ points about this system. You can get them as cheap as 800$, but you have to count in the installationcost if you're not doing it yourself. But DIY is only recommend if you're a professional or have done it before as improper installation could be a fire hazard.
Central AC Units

These ACs are really powerful and normally used to cool entire houses. Thats why they can be quite expensive and use lots of power. If you buy one of these and use them all the time, you should think about buying a solar panel as this would pay for itself pretty quickly. This model is mostly used when already preinstalled in a large grow or a housingcomplex You will need a licensed electrician for this installation as this is really complicated and easily done wrong, which again poses a fire risk
How to setup a coldroom for good workflow
Now that we set up our basic shell for the coldroom we need to bring in our equipment. If we want proper workflow for e.g. commercial use (most hash/time) we have to think strategically on how to setup everything in an order where you don't block others from doing their part. So in order to give you an idea what one could look like, we depicted a example in here

If you don't understand all the terms, no problem, we will now take a look at the step by step tutorial. This should clear up most of the confusion
A step-by-step approach to washing Ice-water-Hash
So we have our room prepared, material sourced and ourselves clothed accordingly. Let's beginn the best part, the wash It's advisable to really check if you have everything you need in the room as opening it again could introduce more contaminants in your room Have the icecubes stored in the freezer aswell so that you can directly take more if you need it
Our material should either be frozen or dried and ready. First we prepare the material by breaking down larger nugs in thumb sized pieces in order to get the optimal surface area while not making them so small that they get grinded up between the ice. Be really gentle with the buds as every handling "destroys" trichomes and with that, reduces your yield.

When this step is finished, we continue to loading up the wash vessel. First put in the workbag, then a thick layer of ice to cool right off and keep the material from being scrubbed against the bottom of the vessel as we're trying to not damage the buds too much. Now put in a layer of material in (a bit thinner) and then a thin layer of ice again. Repeat this till all your material is in
After this, it's time to get it soaked. Use a gentle setting on your water hose and distribute it evenly over the mix until everything is floating. You want the right ratio of ice/water in order to keep the mix cold but not grind the material between ice cubes. Again, the water vortex is whats separating the trichomes from the flower. The best indicator is listening when you stir it up. Maybe do a testrun beforehand without material to get the mix right. There shouldn't be any crushing noises instead only a light collision sound of single ice cubes hitting the side of your vessel. For a example of this sound and maybe one of the greatest videotutorials out there visit Frenchy Cannolis Youtube channel for his course on hash

Let the material sit for 5-7min for WPFF and 10-12min for dried flower in the water to resoak. We do this to let the flower get more flexible so that it doesn't release to many contaminants when agitated.
Then we beginn with the stirring process. Remember to be gentle to the material, but stirr it thoroughly to get everything nicely mixed. You can start with clockwise stirring or a canoeing like move. Continue this for 5-10minutes depending on how golden the water is and how many washes you want to do. If you want to get everything out in one wash and don't care too much about getting the cleanest of the clean fullmelt you can go long 10-15min. This method is quite time saving if you only go for rosin. If you want to separate every grade of hash you go for 5-10min depending on the strain/stiring strength.
We stop stirring and let the trichomes settle for 2-3 min again so that we can skim of water from the top. We do this as it would be really heavy to empty out the vessel in the drainvessel if it's completely filled. If you're running a maschine you only set the timer and come back to open the drainvalve on the bottom to release the water into the drainvessel. I would advise you to take your sprayer/hose and spray down the drained leftovers again as a lot of hash gets stuck between cubes/material if you only drain it from the bottom. Now that we have our bags "filled" we spray down every bag down like this to get contaminants and contaminants pushed through. We continue this with every bags because trichomes that are the right size don't fall through if they're in the right bag (micron = trichome-diameter) and the contaminants fall through to the last bags if you did it right. How to that properly we will watch a video of eldaggy of him spraying down his hash
https://www.instagram.com/p/B-P_mojBYwE/?utm_source=ig_web_copy_link
Afterwards we can scoop the hash from the bags with our spoon that we put in the freezer earlier. It's advisable to cool 4-5 spoons as they warm up pretty quick again. Now depending on our dry method we put the hash on our trays and spread it evenly to not get any water pockets. If we freeze dry we can put it aside/directly in the dryer. If we air dry we need to squeeze out most of the liquid with a 25micron screen, cover the hash and put it in the freezer in order to microplane/sieve it later on. We can repeat these steps until no more hash comes out or we don't like the work/quality ratio anymore and use the rest for blasting/alc extraktion
How to dry wet Hash
As we can't smoke wet hash we have to dry it now. And as said before we need to decide between air/freeze dry as a home hash maker. For commercial washing you can't use airdry as the risk of productloss and the dry time will make it unprofitable. I mean you can do it, but in order to make enough money of that you need brandawarenes.
Whats a Freeze Dryer
This lovely device is also called a lyophilization ofen and dries your hash in a short time. It does this by freezing it down to about -40°C and then slightly heating the trays with the hash, causing the remaining moisture to directly sublimate into a gas. This effect is encouraged by a vacuum that you pull on the drying chamber. This leaves the highest possible content of terpenes in your hash and prevents it from oxidizing which would darken your hash aswell. For info on how exactly this works and how a freeze dryer looks like we will watch a short video from a big drying manufacturer
For the tech heads under you, we have a second option to buying a prebuilt unit. There is the possibility of building your own freeze dryer. This will safe you a lot of money, but beware, it won't give you the same results that a commercial, specialized unit will give you. Here's a short video tutorial
How do I choose and use a Freeze Dryer
Let's assume you want to buy one of these babies but don't know where to look. We would recommend different models depending on your capacity needs and budget. First we have the harvest right models, these are what most people use in the industry as they beginn at 2500€ and a capacity of 800gs of Hash each day of usage. The only problem is the customer service and life time of these maschine as they require intensive care especially with an Oil pump. The second brand are labconco dryers. They get used in the pharma space and are the Ferrari under freeze dryers. These units are built for a lifetime, but also really expensive as they begin at around 15000€. We would recommend them only for commercial users as they won't make sense for the home user.
-

Freeze dryer Harvest Right vs Labconco
But then there is another decision to make. Do I go for the cheaper oil vacuum pump or do I invest in an oilless version
This mostly depends on your budget as I would advise you to always go with the oilless version as this is way less work. You would have to regularly check and change your oil and if it malfunctions you get oil all over your hash. But the oilless version also costs an extra 2000-3000€ depending on the manufacturer.
This brings us to our next point: how do I use my freeze dryer? We already told you about the pump maintenance, but this is not the only thing you have to check before using your dryer. Before each cycle you should check the drain valves. Let out all the drainwater and the close it again tightly, because if you forget that you won't be able to produce a full vacuum. Now we clean the inside of the dryer and remove any leftover ice (if you haven't defrosted). We connect the pump to the dryer and inspect the doors if they fit snuggly on the sealing part that guarantees the ability to pull a vacuum. If we prepared our trays right and they look like this we can load them into our FD and start the process. This will take about 24-36h depending on how thick your hash layer is

To check if it's dry, take a card and move the hash around a bit. If it behaves like sand and has no clumps left it is dry and ready for smoking or further processing
Drying Hash without a freeze dryer
This method is mostly used for home made hash for it's cheaper tools. As said in the washing guide you should immediately freeze your hash after collecting until it's a hard brick. This gives you the opportunity to separate the individual trichomes better in order to get more surface area for water to evaporate. You need a temperature controlled room with about 30-40% humidity at all times! The room should also be clean to insure no mold spores get into your hash and multiply.

Now you take baking paper sheets and lay out your drying area. You can use baking trays in an open room setup or put it in a clean (!) pizza Box as the cardboard will pull out even more moisture. Then take your frozen hash bricks and grate them either with a microplane or a siever finely over the paper. Be careful to really spread it out good as clumps may lead to inconsistent drying. Wait approximately 1-2 weeks and check regularly for dryness.
How do I calculate my yield from flower to Hash
Now that we weighted out dry hash and have a g number we can simply take the % Value e.g. 5g Hash out of 100gs Flower would equal a yield of 5% These values differ greatly between WPFF and cured Hash as you have to subtract the water weight from the fresh frozen flower. The fresh flowers are about 75-80% water so 5% is a high yield compared to cured hash yield where 20-25% would be a good yield. Good fresh frozen yields are between 3-8% depending on the strain, 8% being the top of the top. Most times these numbers are only achieved with selected cuts asa lot of strains yield <1% For cured Hash a good range is between 15-30% return, but this also requieres a hash specific cut
More Hash-Education
If you want to learn more about Hashtech and Hash culture visit my buddy over at TheHashishInn and listen to his Podcast

What does colour say about my Water-Hash
This is a very controversial discussion in the water-hash space and I hope to not step on anyones to toes, but this needs to be said. Colour doesn't affect the quality of Hash! Most use this phrase but forget to say that, while this is true, there can be bad dark hash. Sometimes it's not a sun ripened or cured resin, but just contaminants. This is a important factor to consider when buying Hash. You have to look really close and do the ultimate test: how does it smoke! No real hash connoisseur cares about the colour when the flavor, smoothness and high is right and so should you. Some people go as far as harvesting way to early in order to get the whitest hash, while it only leads to a speedy, weak high and nearly no flavor
"Dark" or better said amber hash can come from strong UV-Radiation, CBN-Content or long air exposure which oxidizes the compounds in hash
How is Hash rated?
Most of you will have heard about 6 star hash or lower quality, but exactly is that measured? Tbh the star-rating system is used quite liberally so a lot of "six-star" hash is worse or not even close to being fullmelt. To give you a quick overview of how it should be used and a measurable alternative shall be given here
The star rating system
1-2 Star
This grade is mostly used for edibles/RSO or further cleaning processes as it's from low quality. You can identify it by it's green/dark colour that stems from the high content of contaminants. You will typically find this in the 180u or 25u bags
3-4 Star
We use this grade of water-hash for edibles on the worse end and on the better end of the quality spectrum we press it to rosin as this will still yield a proper, terpy oleo-resin. Typically found in the 150u Bags and characterized by it's light colour but missing meltyness
5-6 Star
This premium product will be processed to high-end-rosin on the lower quality spectrum and the best of the best will be left as it is if you have a market for melt. If not, you can also press it and sell it for the maximum rosin price, but beware I will call it a sacrilege to press such high quality melt haha True fullmelt shouldn't leave residue in your banger. A tiny bit is normal as the trichome heads still have their waxy cuticle and this "burns"
The gravimetric measurement, a good alternativ?
As you might have noticed the star-rating leaves a lot of room for interpretation and therefore is unprecise. Fletcher from Archive Genetics wanted to adress this issue and bring in a measurable test. As he was the one to bring freeze dryers into the hash industry, he has a lot of credibility in this regard.
His method is to use an enail, take a 15micron metal screen and weight it before. Now you put exactly 0,1g of your Hash on the screen and put all that on the enail until no oil is left. Then take the screen and weigh it again. Now subtract the previously taken screen weight and there you have your residueweight. This also can be converted into a %melt value which should be used to categorize the Hash Visit his Instagram for fire flower and melts
Sieh dir diesen Beitrag auf Instagram an
Ein Beitrag geteilt von Archive Seed Bank (@archiveseedbank) am Apr 19, 2020 um 6:09 PDT
What can I do with my finished product?
This depends on your needs, but most people dab/press it or use it for high end edibles. If you process it to rosin you can also make carts with it or fill preroll "donuts" with it as this is quite popular right now The leftover washed flower can be freeze dried and be blasted/alc washed to get every cent out of it. If you already separated all trichs you can use it as mulch for your garden aswell. I hope you learned something new and had fun doing it. If you have any tips/tricks or corrections you want to be included, feel free to leave a comment or shoot me an email as I want to improve the quality of my hash making aswell and you never stop learning Happy dabbing everyone PS: Sorry for my english, I'm not a native speaker :D
Disclaimer: This tutorial is intended for legal use in legal states only. We do not condone any illegal activity
Sources
- Dr. Ethan B Russo; "Taming THC: potential cannabis synergy and phytocannabinoid-terpenoid entourage effects"; (https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3165946/)
- Analytical 360; „Bubble Gum Hash Rosin“ 16.06.2016 http://archive.analytical360.com/m/concentrates/565901I
- Iceextract; ICExtract Bag Singles and Kits 5Gal V3 https://icextract.com/icextract-bag-singles-and-kits-5gal-v3/
- Pollonator/Mila Jansen; MEDIUM ICE-O-LATOR® 7 BAG SET; https://pollinator.nl/product/medium-ice-o-lator-7-bag-set/
- GoPurePressure; Bubble Now 220 Micron Work Bag https://gopurepressure.com/collections/bubble-bags-machines-bubblehash/products/bubble-hash-bubble-now-220-micron-work-bag
- homeairguides.com; How To Choose An Air Conditioner (Step-By-Step Guide On Picking The Right AC Unit; https://homeairguides.com/air-cooling/how-to-choose-an-air-conditioner-guide-on-picking-the-right-ac-unit/
- Deborah Hucht; Kühlleistung berechnen: Welche Klimaanlage eignet sich für welche Raumgröße?; Kühlleistung berechnen: Welche Klimaanlage eignet sich für welche Raumgröße?
- Ab Hanna; "Hash Rosin 101: Lessons from Experienced Solventless Extractors"; https://hightimes.com/guides/hash-rosin-101-lessons-from-experienced-solventless-extractors/
- PurePressure; Andrew Ward; "How Bubble Hash is Rated (1* to 6*)"; https://gopurepressure.com/blogs/rosin-education/how-bubble-hash-is-rated-1-to-6
- Titelbild: CubanGrower/https://www.instagram.com/cubangrower/ / https://www.instagram.com/_thevillage/
- "HOW DO REVERSE OSMOSIS SYSTEMS WORK?"; (https://kraaiwelldrilling.com/how-do-reverse-osmosis-systems-work/)
- "Different Types of Terpenes in Cannabis"; (https://tetrahorizon.com/2020/07/10/different-types-of-terpenes-in-cannabis/), TheProfessor
Plant Tissue Culture - the future of Cannabis clone production
featured image by the one and only @kandidkush
1. Introduction
In recent years, automation and standardization of the crop production process are becoming increasingly important. Especially in the production of cannabis, it is crucial to have uniform and disease-free planting material in order to reduce any plant protection measures to a minimum and to ensure a final product with consistent quality. For this reason, more and more growers decide to source their seedlings from in-vitro propagated or tissue cultured material. But what does In-Vitro production and tissue culture even mean? And how is it done with cannabis? That is what we want to explain in this Article.
In Vitro is Latin and means “in glass” or “in the glass” therefor when according to plant production, it is a technique that uses glass containers such as petri dishes and test tubes as a controlled artificial environment for the propagation of plantlets. This contrasts with in-vivo ("within the living") and in-situ ("on site") production, which are commonly used in horticulture.
Within in-vitro production there are various methods and ways to propagate the desired type of explant. Tissue culture and micropropagation are two terms that you will most likely come across while looking closer into that topic. But what exactly is the difference between those?
The main difference between micropropagation and tissue culture is that micropropagation is the production of a large number of plants from a small amount of plant material, whereas tissue culture is the first step of micropropagation, where plant cells are grown in an artificial medium to develop them into a large number of plantlets. In addition, micropropagation requires tissue culture for the propagation of plantlets.
As the global market advances to a greater reliance on plants for active ingredients, delivery of consistent and pathogen free plant material is crucial. Anyways, in the production of medicinal or recreational cannabis, the use of these techniques is not yet widespread, as cuttings can also be produced via "normal" vegetative propagation but, the higher the degree of automation and the producers' demands for cleanliness become, the more this method comes into focus. In addition to the possibility of producing clean plants, in-vitro culture is also suitable for space-efficient growing, improved yields due to vigorous plants and for saving production costs.
However, the in vitro cultivation of plants is not only suitable for propagation but is also of great interest for research and especially for breeding. For example, pathogens can be eliminated from plants, mutations can be created and sterile backups of rare or hard-to-retain plants can be secured. There are many ways to utilize tissue culture and other biotechnological methods and there are even more to be discovered.
2. Basic Terms for In-Vitro production
2.1 Medium
2.1.1. What's Agar Agar?
Some of you might know it as a plant derived gelatin substitute and that's nearly the same purpose we will use it for. The thickener is based on a galactose containing extract taken from red-purple marine algae. This brings the typical nutrient solution to the right consistency that supports vigorous growth, but gets easily penetrated by the roots to insure the best development possible. In order to generate the perfect Agar mixture we have to add it in a ratio (depending on the source) 1-2:100. This leads to a nearly complete solid medium. If you want to cultivate in anaerobe/liquid conditions, you can simply half the concentration.

´
But why not just use gelatin instead?
That's a good question, but there's a simple answer to it. It's edible to bacteria and most other microbes so you would have to refill every plate after a short time. The nutrient dosing and validity of the experiment undertaken would be in danger as you would have to add the gelatin to the feeding chart of the microbes that are cultured.
It comes in different forms, but the most common are flakes, that need to be mixed with water und constant heat application until it's fully homogenized. These flakes are easy to store and quite cheap to get. If you have the necessary budget, there are pre-mixed/pre-sterilised versions, mostly bottled up, that can be used instantly without waiting for the hot agar mix to cool down. The cooling process can take up to 30min per batch before use.
Not only the distribution form differs but also the additives. Many agar mixes have added ingredients for certain purposes as this is used for all types of cell cultures from human cells to the infectious disease causing Escherichia coli.
The potato-dextrose agar for example is the most used form for fungi especially for varieties like Botrytis cinerea also known as the common grey mold. Another one is blood agar, which as the name already says, contains animal blood in order to study special microbes. For tissue culture the most common type is the standard galactose agar in combination with a nutrient matrix and a hormone, depending on the phase.

The standard nutrient matrix in nearly all cell culture work is the Murashige-Skoog-Medium, which contains simple amino acids, nutrient salts and photosynthesis products (saccharose) as the plant can't photosynthesis in most cases. Inositol is also added in order to mitigate plant stress, strengthen cell walls and phosphate storage.
2.2 Hormones for plant tissue culture - is that even safe?
Many myths rank around plant hormones, which are mostly "feared" by the Cannabis community as it only has found real application in large scale agriculture. They get mixed up and thrown in the same bucket as GMO and Glyphosate. Not that those would be bad substances but the social stigma around them is strong.
The hormones on the other side are nearly identically to their natural/plant counterparts, which control nearly all the physiological responses of the plant to outside influences like the movement of the sun or the alignment of the plant to the gravity
2.2.1 Auxin
The first of these phytohormones (hormones in plants) is responsible for a multitude of responses to environmental stresses. Most auxin derivates in the plant and made by humans are based on the Indol-3- acetic acid. This compound is mostly synthesized in the apical region refering to the main growingtip of the plant. From there on it gets transported via the phloem and cell-to-cell by the PIN 1-9 transporterprotein.
The basic effects include wound response, apical dominance (the main branch is the tallest one), photo-/gravitropisms and it plays a important role in fruit development. But lets dive a bit deeper in the interaction of Auxin and the use cases of it.
The first practical application would be induction and promotion of root growth. This is the most used purpose and nearly all commercial rooting gels contain it. But not only in Cannabis, but also the floral industry uses it predominantly in their rooting SOP.
We also use this by giving our explants (plants in in-vitro conditions) a certain amount of it in order to achieve the highest rooting rate possible.
But as with everything in life, there is a limit to whats good for the plant. Excessive application of this substance can result in growth inhibition and plant death. Weedkillers like 2-4-D are based on the synthetic counterparts of auxin like indole-3-butyric acid. But not only damaging substances also "good" ones like common rooting gels are based on synthetic auxin.
"Natural" Auxin can be found in variety of plants shoots especially the ones of the willow tree.

2.2.2 Cytokinin
The next hormone in our repertoire is the counterpart to the previously mentioned Auxin. This compound focusses on the lateral growth of both roots and shoots. To be more exact the ratio of Cytokinin:Auxin is the driving factor in the morphology of the plant. They not only contradict each other, but also work together when influencing cells. When only Auxin is applied cells elongate and become big, but won't expand or differentiate. Same takes place when the ratio is 1:1. This is used for Callus multiplication or expansion.
Complementary to Auxin it is synthesized in the roots and travels to the shoots via Symplast and Apoplast. Production of this chemical is regulated by two types of response regulators. One being the B-Type and the other being the A-Type. Both are regarded as transcription factors as the either activate or stop the production of Cytokinin via influencing the transcription of the corresponding gene loci.

Using this knowledge we can manage the morphology of the plant into growing more squat thus increasing internodal stacking and use of space.
But not only the space can be used more efficiently also the time saved by breaking seed dormancy can save time when germinating seeds for a phenohunt. The dormancy is induced by abscisic acid and by increasing metabolic activity the cytokinin decreases the level of abscisic acid in the seed. It's commonly used in plants that have a hard time germinating.
The next effect might be especially interesting for the photographers and florist under you. Cytokinins can delay senescence in other words the decay of the plants. This is achieved by increasing synthesis and slowing decay of certain proteins. Also nutrients are drawn into the treated area from nearby tissue. It's suspected that an enzyme is responsible for these actions, but no scientific consensus has been reached yet.
When using Cytokinins you have to differentiate between the plants own, adenine based, Cytokinins like Kinetin and Zeatin. Others have been found outside of plants, but they are based on Phenylurea. These exceed the effectivness of the Cytokinin in certain plants. Examples for this variety are TDZ and Diphenylurea, which are widely applied in agriculture
2.2.3 Gibberellic Acid
One of the most interesting phytohormones is the counterpart to abscisic acid. Exogenous GA3 (short version) is also responsible for breaking dormancy where it activates indigenous synthesis of more GA3, which adds to dormancy breaking factors. These are hypothesised to be a combination of growth promoting hormones (GA3, Cytokinin, Auxin etc) and reduced nutrient storage in the endosperm. Enzymes, primarily α-amylase, lead to the processing of sugars and other storage units. Another restricting factor is the Cuticula strength of the seed.
The application on an adult plant leads to a big stretch in all shoots. This is principle is also used in plant tissue culture and for virus cleansing. But this has to be done with the utmost focus on the concentration of GA3 as a high amount leads to different sex expression. This mechanic is commonly used in feminized seed breeding as it "reverses" a female plant into producing pollen with female Genome. In even bigger concentration it can lead to the complete sterilization of the specimen. Most fruit growers like in the citrus industry use the mechanic to suppress undesirable seed development
That said, when a trained horticulturist with a strict, thought through regime applies GA3 in flower, it can increase inflorescence mass and trichome development. For homegrowers and people that don't have access to these chemicals, Kelp/Seaweedextract is a perfect allrounder as it contains a variety of growth hormones aswell as Macro-/Micronutrients.
The compound was discovered by japanese scientist while researching the Gibberella fujikuroi fungi. Cytokinin is a secondary metabolite to the pathogenic pathway of the fungi on rice plants.
2.3 Basic Tools for Tissue Culture Propagation
2.3.1 Laminar Flow Hood
It's one of the most important, if not the most important part of a Culturelab. The Laminar Flow Hood (LFH) insures that all steps are done in a steril environment. There is a differentiation between normal Flow hoods and special models for working with microbiological organisms. The first doesn't filter the incoming and outgoing air, leaving the worker exposed to aerosols and microorganisms.
That's why we use the second model in most cases. This LFH got a specialized HEPA-Filter before the air enters the Hood. After getting in it can be distributed in two ways. Vertical flow hoods and horizontal flow hoods can be differentiated easily as word explains itself
All work including the plant have to be done inside to keep out potential contaminants. It's therefore of vital importance to wipe down all areas inside with ethanol (70%) and clean the outside of culture vessels aswell when introduced in the steril environment. Gloves and hair-protectants are a must, if mass-propagation is planed. At home you can easily get away with disinfecting your hands and arms up to the elbow. Another concern is your breath as it often contains bread-mold spores and a variety of microorganisms that thrive on tissue culture medium.
If a infection takes place and you have no copy/other option, you can add antibiotics to the medium, but be very careful. Most higher plants are more resistant to these compounds than the microorganisms, but the optimal balance is very hard to reach. Another factor is the difference between every plant genome/phenotype thus a cultivar-specific test would be necessary before adding antibiotics.
2.3.2 Autoclave
Speaking of sterilisation, we will subsequently be lead to autoclavs. These laboratory certified pressure cookers are an essential tool for cleaning glassware and used culture vessels. They come in different sizes and forms, which you can choose specific for your application.
Despite the different shapes, the mechanism is the same. The chamber is evacuated via a Vacuum pump in order to simplify the sterilisation progress. It gets filled with hot steam and thus put under a high pressure to increase the cleaning efficiency. After 15-20min the optimal cleansing effect is achieved, which means that the content can be removed. Be careful not to touch it with your bare hands as it's still really hot.
You should also size the autoclave big enough for the operation, but not too big for your batchsizes as this will lead to unnecessary cost for energy.

2.3.3 Magnetic Stirrer
This device is rather simple and doesn't need much explanation, but it's still vitally important that we talk about it. You probably wonder why and we will will explain it shortly. Not the stirrer per se needs explanation, but rather how to use it and what for. It's the base of a good tissue culture lab as it's used for mixing and homogenizing of the medium components that we discussed earlier.
Each component has a specific boiling/inactivation point that you have to consider when mixing.
The goal is to find the right compromise between solubilityspeed and retention of effectiveness. Some compounds like hormones derivates have to be added after the mixing + autoclaving of the medium as they can't even withstand the autoclaving procedure.
For these substances you have to consider other ways of sterilising them such as syringe filters or specific solvents.

It enables the production of large amounts of media as it's fully adjustable in rotationspeed and temperature, if you have a heated version. Be careful to pick a appropriable sized stir bar as this determines the homogeny of your mixed substance. But even with a large stir bar the stirrer has it's limitations when the mixing materials are too viscous. If that's the case, we would advise a mechanical alternative as they can plow through it better. Keeping that in mind, you have to pour your hot, mixed medium out of the vessel before it cools down as it gets really hard to remove the bar after.
2.3.3 Consumables
This chapter is for items that are important, but didn't get a separate chapter as it would go beyond the scope of this article.
Firstly we have your PPE like gloves, masks, Labcoats and hair nets. These are especially important, if you have a large team and thus many vectors of contaminants/pests. But keep in mind that PPE doesn't eliminate the necessity of disinfecting the whole work environment before and after you do explants in it.
Another vital point are the culture vessels. These are the new homes for your little clones and need to have a large enough volume to accommodate them. This refers especially to the amount of medium that a plant needs in its stage of growth. There are mayor differences from cultivar to cultivar so the best way to go about it, is to experiment with it and keep tight journals in order to create SOPs. This takes a lot of time, energy and labour so be mindful of that, when trying to establish a plant tissue culture production.
Our last important item is the scalpel. You can find many different forms of it for specific applications, but we recommend that you go for a more environmentally friendly version by using a scalpel holder with exchangeable blade. Using this will decrease your waste of material and money as the amount of cut plant material in an TC production puts a great stress on the blade thus making it dull. The alternatives would be to use whole single use scalpels, but the produce large quantities of plastic that we don't want or high end versions. The later stays sharp for longer, but not long enough to make up for the huge price difference.
2.4 Standard terms for plant meristem tissue culture
In order to understand the basic mechanics of saving genetic material and growing callus cultures we have to take a look at the structure of a growing plant tip. This is the location we're using to explant a meristem from. But what is a meristem?
The term comes from the ancient greek word merisein, which means divide. It was used by the swiss scientist Carl Wilhelm von Nägeli to describe the undifferentiated, multiplying Cells that made up the growing tips of the plant. The apical meristem is the highest growing tip in the plant and sits on top. The counterpart of this region is the basal meristem, which sits of the tip of the roots as you might have guessed.

Photomicrograph of a Coleus stem tip. A=Procambium, B=Ground meristem, C=Leaf gap, D=Trichome, E=Apical meristem, F=Developing leaf primordia, G=Leaf Primordium, H=Axillary bud, I=Developing vascular tissue. Scale=0.2mm.
The plant builds new meristemcells in the top middle parts as the lower parts differentiate into the predetermined functional parts. Some go to be Parchenym cells, some are going to be part of the vascular system of the plant. Knowing this, we can extract these cells before they're differentiated in order to get totipotent cells (cells that can be any part of the plant). These are full of potential and are kept in a cryofreezer to be saved for later usage, as backup or as basis for mass cell production in a bioreactor.
3. Basic mechanics of mass micropropagation
To get an idea of how micropropagation could benefit your growing operation we're gonna draw a little plan of how it could look like.
First of all we need a small motherstock from which we can take our microcuttings. These are about 2,5cm/1 inch long and at best, taken from a meristematic region.
Then we plant it in a multiplication medium under sterile conditions in order to avoid contamination and the subsequent demise of our plant matter. After 2-3 weeks we should see 2-4 new shoots coming from our tissue, which we can also cut up into separate tissue samples. The we put them in individual containers with the same medium mix again and repeat the previously mentioned cycle. Even thought there will be loses due to contamination, the replication-rate is extremely high due to each new tissue replicates itself 2-4 times. If we calculate this through, we can see that with minimal space use, an enormous plantcount can be achieved in a short period of time.
After we finished the replication cycle, we can move onto the rooting phase. This uses a special medium containing auxin derivates, which induce the production of roots on our previously made shoots. After 2-4 weeks again, we have to inspect the plants accordingly and if they're sufficiently developed, we beginn the hardening phase.
For this part of the productioncycle the plants get translated in the medium of choice that will be used later on in production. Most growers will use rockwool as it has excellent water holding capacities, but also coco or small soil pots can be used. Light plays a huge role now and has to be increased in order to acclimate the plants to the later vegetative conditions.
The humidity also has to go from the comfy 80-90% RH in the boxes down to the values of your vegroom.
This was a quick overview of the process and as you could see, there are many obstacles along the way, but in the end it's not that hard to achieve at home. For this exact purpose we will compare DIY kits that can be bought online with a home made microlab.
4. What are ready-made home kits?
There are some suppliers on the internet who offer hobby kits for micropropagation. Even if these kits are suitable for experimenting and for first experiences, they cannot replace a professional laboratory, because similar to mushroom cultivation, a sterile working method and environment must be guaranteed to avoid failures. In addition to all the materials and equipment, professional application requires trained personnel and constant quality control of the work process.
5. Future vision
As cannabis production becomes more and more automated and the costs of labor, electricity and rent continue to rise, we at research gardens see in-vitro production as a key advantage for companies that want to remain sustainable in a rapidly evolving industry. Due to the prospect of a long-term reduction in production costs as well as a simultaneous increase in product quality your company will get an advantage against your competitors. Particularly in pharmaceutical operations, a consistent quality of the plants and ingredients is indispensable. For the recreational market, however, this is just as important since similar requirements exist here as well.
Interested? Then let us start working out a concept for an in-vitro laboratory in your company today! Send us an e-mail to info@research-gardens.com
Sources:
- "Communication by Plant Growth Regulators in Roots and Shoots of Horticultural Crops" by Anish Mallad; Jacqueline K. Burns; August 2007; HortScience: a publication of the American Society for Horticultural Science 42(5) (https://www.researchgate.net/publication/237379591_Communication_by_Plant_Growth_Regulators_in_Roots_and_Shoots_of_Horticultural_Crops/figures?lo=1)
- "Hot Plate with Magnetic Stirring: 6.7"x6.7" SS Plate Max.300C - SH3"; MTI Cooperation; (https://www.mtixtl.com/hotplateswithmagnetcstirring67x67stainlesssteelsurfaceupto300c-eq-sh-3.aspx)
- "Sterilization cycle phases for a steam sterilizer"; rsd-engineering.com; (https://www.rsd-engineering.com/steam-sterilization-cycles)
- "Laminar flow hood/cabinet- definition, parts, principle, types, uses"; by Anupama Sapkota; (https://microbenotes.com/laminar-flow-hood/)
- "Baum HD"; Dreamliner (https://wall.alphacoders.com/big.php?i=612687&lang=German)
- "CULTURE MEDIA AND ITS TYPES USED UN MICROBIOLOGY LAB."; milan; (http://biologyofmicroorganisms.blogspot.com/2010/09/culture-media-and-its-types-used-un.html)
-
"[Tuesday Scoop] Puzzlement Between Agar-Agar And Gelatin"; Namrataa Mahalley; (https://bulbandkey.com/blog/tuesday-scoop/puzzlement-between-agar-agar-and-gelatin/)